Vascular Intervention // Coronary
Resorbable Magnesium Scaffold (RMS)
Magmaris

 Compelling safety data
Compelling safety data
 Fast Magnesium resorption time
Fast Magnesium resorption time
 Better deliverability
Better deliverability

Why Magnesium?
Magnesium alloy: Favorable mechanical properties of a robust Magnesium backbone
Robust Magnesium backbone
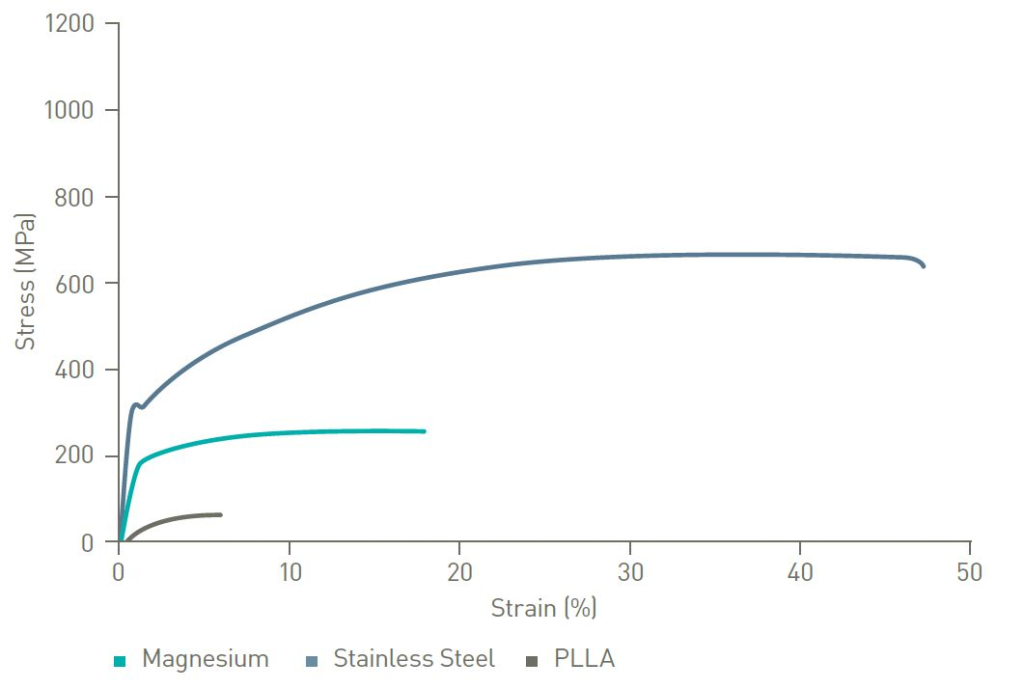
The mechanical strength of Magnesium is superior to polymers like PLLA.1
Stable recoil
Magmaris has a 38% lower recoil after 1 hour.2
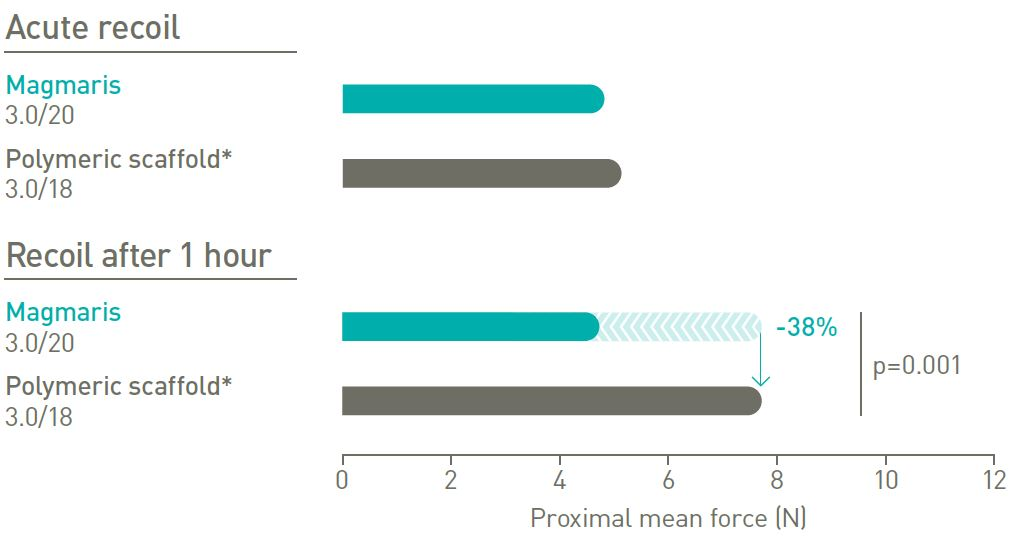
*Absorb, Abbott
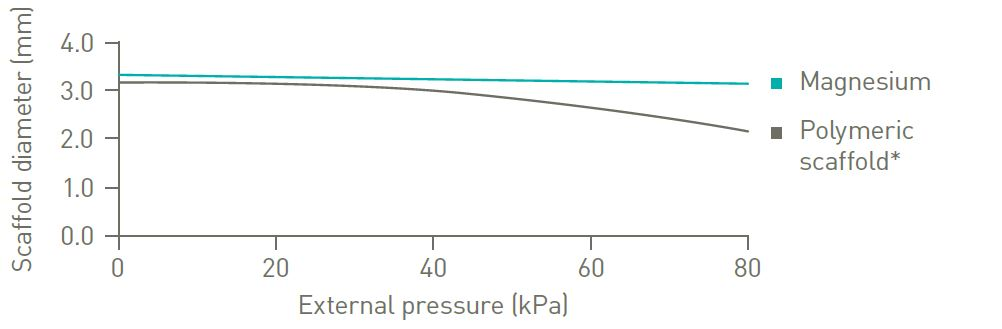
Strong radial resistance
No significant diameter change under increasing physiological pressure.3
Rounded edges and smooth surface
The electropolished rounded edges and smooth surface of the Magmaris scaffold generate less resistance during delivery of the scaffold to the lesion.


Compelling safety data
Confidence through evidence
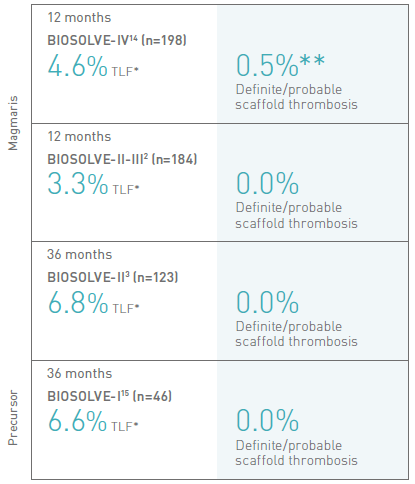
*Target Lesion Failure. Composite of cardiac and unknown death, target vessel myocardial infarction, clinically driven target lesion revascularization and CABG.
** Patient underwent MIDCAB with subsequent DAPT interruption 5 days after the procedure.

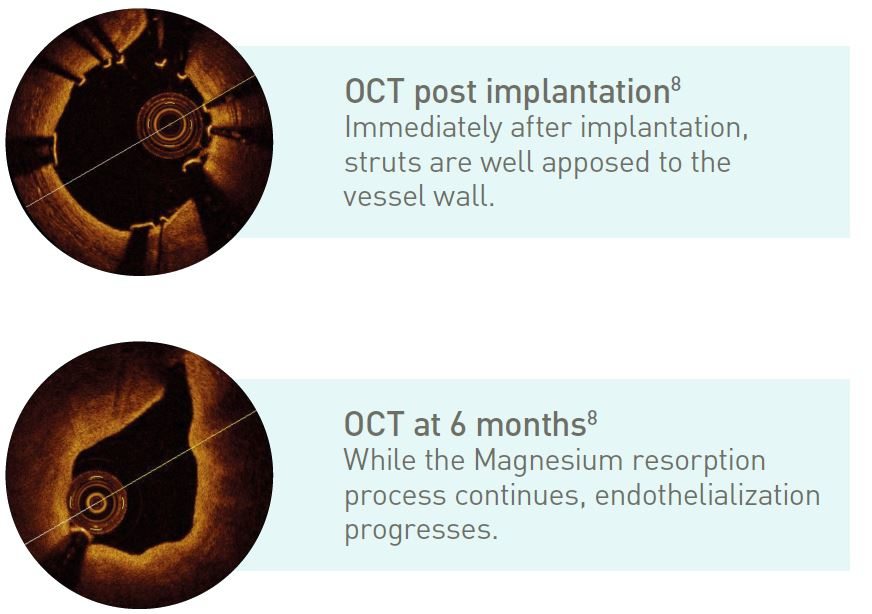
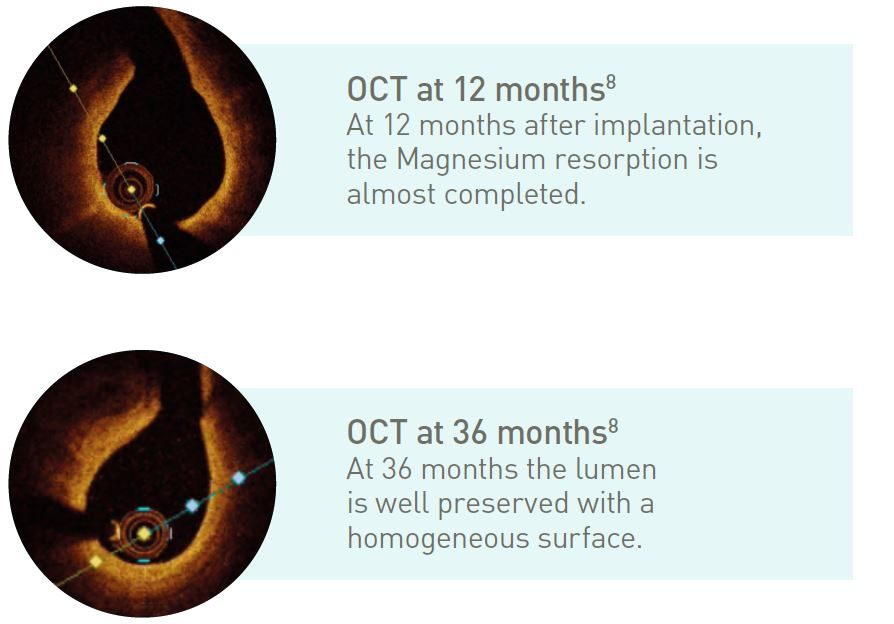
Fast resorption time
~95% of Magnesium resorbed at 12 months7
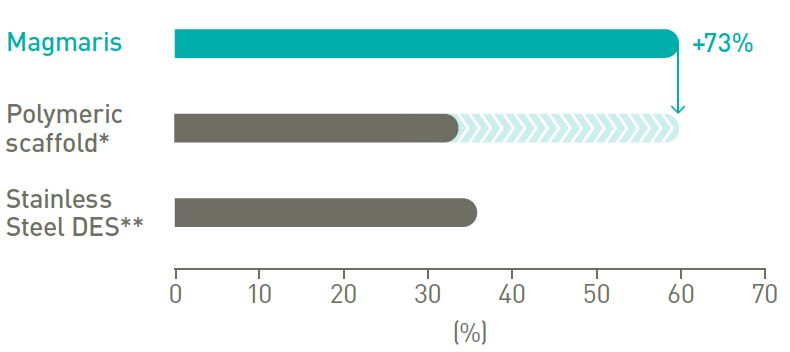
A more deliverable scaffold
More than 70% of physicians who have used Magmaris RMS in clinical practice have rated the device to be better than a polymeric scaffold.9*
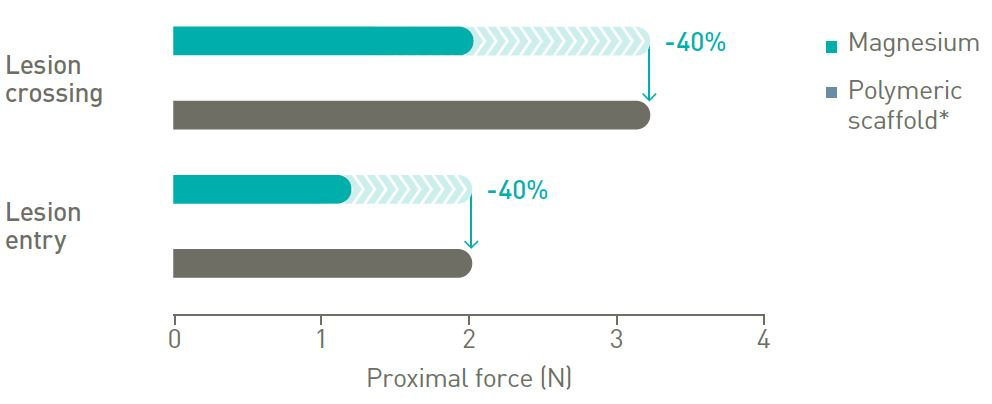
Better lesion crossing
Up to 40% lower lesion entry and crossing force.10
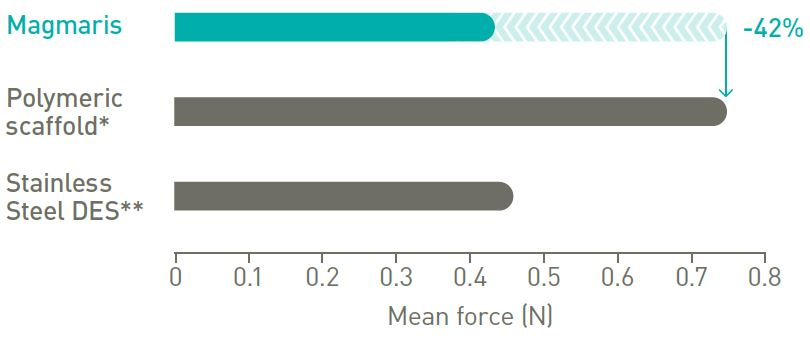
Better trackability in tortuous anatomy
42% less peak force.11

Better pushability
73% more force transmitted from hub to tip.12
* Absorb, Abbott
** BioFreedom, Biosensors

 Magmaris
Magmaris
Indicated for de novo coronary artery lesions.*
Technical Data
| Scaffold | |
|---|---|
| Scaffold material | Proprietary Magnesium alloy |
| Markers | Two tantalum markers at each end |
| Active coating | BIOlute bioabsorbable Poly-L-Lactide (PLLA) eluting a limus drug |
| Drug dose | 1.4 μg/mm2 |
| Strut thickness/width | 150 μm/150 μm |
| Maximum expandable diameter | Nominal Diameter +0.6 mm |
| Delivery System | |
|---|---|
| Catheter type | Rapid exchange |
| Recommended guide catheter | 6F (min. I.D. 0.070") |
| Crossing profile | 1.5 mm |
| Guide wire diameter | 0.014" |
| Usable catheter length | 140 cm |
| Balloon material | Semi-rystalline polymer |
| Coating (distal shaft) | Dual coated |
| Marker bands | Two swaged platinum-iridium markers |
| Proximal shaft diameter | 2.0F |
| Distal shaft diameter | 2.9F |
| Nominal pressure (NP) | 10 atm |
| Rated burst pressure (RBP) | 16 atm |
Compliance Chart
| Balloon Diameter x Length (mm) | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nominal Pressure | atm8 | 10 | 10 | ||||||||||||||||
| (NP) | ø (mm) | 3.00 | 3.54 | ||||||||||||||||
| Rated Burst Pressure | atm8 | 16 | 16 | ||||||||||||||||
| (RBP) | ø (mm) | 3.29 | 3.82 | ||||||||||||||||
Ordering Information
| 3.00 | 412526 | 412527 | 412528 | |||||||||||
| 3.50 | 412529 | 412530 | 412531 | |||||||||||
Contact

1 BIOTRONIK data on file;
2 BIOTRONIK data on file;
3 BIOTRONIK data on file;
4 Waksman R. Safety and Clinical Performance of the Drug Eluting Absorbable Metal Scaffold in the Treatment of Subjects with de Novo Lesions in Native Coronary Arteries at 12-month follow-up- BIOSOLVE-II and BIOSOLVE-III. Presented at tct 2017; October 31, 2017; Denver, USA;
5 Haude M, Ince H, Kische S, et al. Sustained safety and clinical performance of a drug-eluting absorbable metal scaffold up to 24 months: pooled outcomes of BIOSOLVE-II and BIOSOLVE-III. EuroIntervention. 2017;13(4): 432-439;
6 Haude M, Erbel R, Erne, et al. Safety and performance of the Drug-Eluting Absorbable Metal Scaffold (DREAMS) in patients with de novo coronary lesions: 3-year results of the prospective, multicentre, first-in-man BIOSOLVE-I trial. EuroIntervention. 2016; 12(2): e160-6;
7 BIOTRONIK data on file;
8 BIOSOLVE-II case, GER443-001. Courtesy of M. Haude, Lukaskrankenhaus Neuss, Germany 2015.
Absorb is a registered trademark of Abbott Laboratories; BioFreedom is a registered trademark of Biosensors Europe.
*Indication as per IFU.

