BIO|CONTINUE
BIOtronik Study to Assess the CONTINUation of Existing Risk of Ventricular Arrhythmias After CRT-D Replacement for Patients With Primary Prevention Indication
Study Design
- Prospective, observational, international, multicenter study
- Assess the relevance of a defibrillator back-up in CRT-D patients with an ICD indication for primary prevention after the replacement of the first CRT-D
- 276 patients
- ≥ 24 months follow-up after device replacement
Key Result 1
The rate of patients with sustained ventricular arrhythmia events (sVAE) was 8.3%, 10.3%, and 21.2% at 1, 2, and 4 years post replacement.
Image
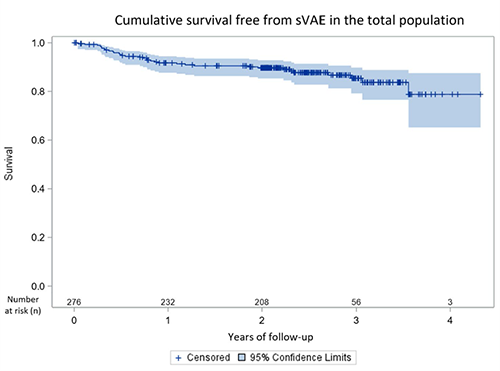
Key Result 2
Patients without persistent ICD indication at replacement still had a sVAE rate of 5.7% (95% CI 2.3–11.5%) at 2 years.
Image
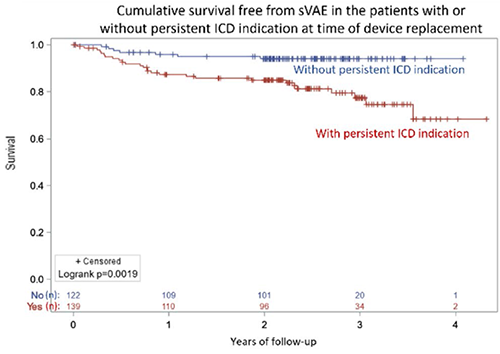
Key Result 3
In multivariate analysis, predictive factors of subsequent sVAE were:
- persistent ICD indication (hazard ratio (HR) 3.6; 95% CI 1.6–8.3; p = 0.003)
- 64–72 years of age as compared to ≥ 79 years (HR 3.7; 95% CI 1.4–9.7; p = 0.008)
- ischemic heart disease (HR 4.4; 95% CI 2.1–9.3; p < 0.0001)
Clinical Relevance
- BioCONTINUE shows a persistent risk of sVAE in patients with and also without persistent ICD indication at the time of device replacement
- The risk of sVAE depends on age, a persistent ICD indication at the time of dervice replacement, and on the presence of an ischemic heart disease
| Study Objective |
|
|---|---|
| Primary endpoint |
|
| Secondary endpoints |
Rate of patients with at least one sustained ventricular arrhythmic event (sVAE) in patients with a
|
| Clinical Sites |
|
| Sample Size |
|
| Inclusion Criteria |
|
| Main Exclusion Criteria |
|
| Devices |
|
| Follow-Up |
|
| Study Duration |
|
| Reference No. |
|
| Principal Investigator |
|
Gras, D., Clémenty, N., Ploux, S. et al. CRT-D replacement strategy: results of the BioCONTINUE study. J Interv Card Electrophysiol (2022). https://doi.org/10.1007/s10840-022-01440-5
