Thinner struts for low Chronic Outward Force (COF)²
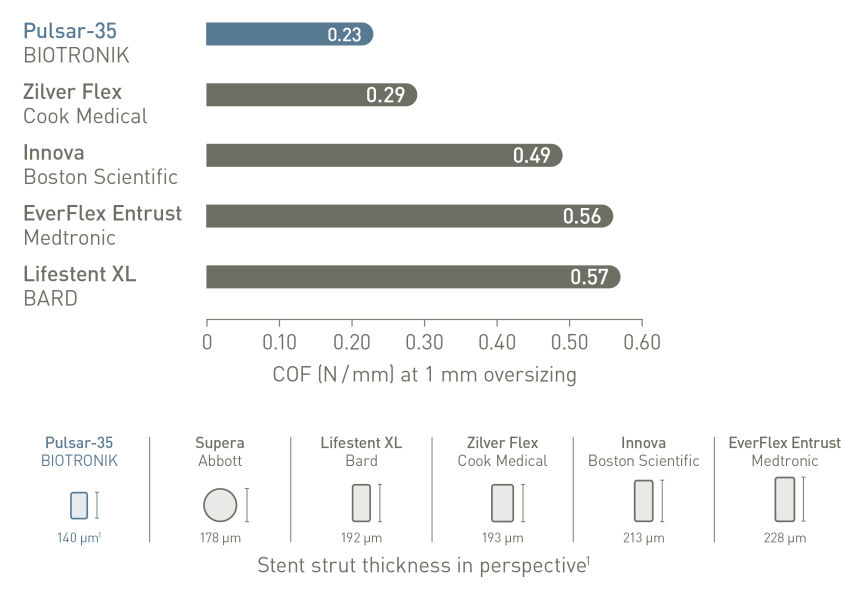
Thinner struts and lower COF make a difference:*
• Lower risk of restenosis3
• Reduced vessel injury and inflammation3
• Faster endothelialization4,5
Indicated for use in patients with atherosclerotic disease of the femoral and proximal popliteal arteries, in particular for the treatment of insufficient results after Percutaneous Transluminal Angioplasty (PTA), e.g. residual stenosis and dissection.*

• Lower risk of restenosis3
• Reduced vessel injury and inflammation3
• Faster endothelialization4,5
¤Clinical outcomes of Pulsar-18 can be used to illustrate clinical outcomes of Pulsar-35 due to identical stent platforms FTLR - Freedom from Target Lesion Revascularization; PP - Primary Patency; A.L.L. - Average Lesion Length
Sufficient radial force for a long term vessel support, even in calcified lesions
| Stent | |
|---|---|
| Catheter type | OTW |
| Recommended guide wire | 0.035” |
| Stent material | Nitinol |
| Strut thickness | 140 μm |
| Strut width | 85 μm |
| Stent coating | proBIO® (Amorphous Silicon Carbide) |
| Stent markers | 6 gold markers each end |
| Sizes | ø 5.0 - 7.0 mm; L: 30 - 200 mm |
| Proximal shaft | 6F, hydrophobic coating |
| Usable length | 90 and 135 cm |
| Stent ø (mm) |
Catheter length 90 cm (Stent length mm) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 30 | 40 | 60 | 80 | 100 | 120 | 150 | 170 | 200 | ||
| 6F | 5 | 379878 | 379879 | 379880 | 379881 | 379917 | 379918 | 379919 | 379920 | 379921 |
| 6 | 379883 | 379884 | 379885 | 379886 | 379922 | 379923 | 379924 | 379925 | 379926 | |
| 7 | 379888 | 379889 | 379890 | 379891 | 379927 | 379928 | 379929 | 379930 | 379931 | |
| Stent ø (mm) |
Catheter length 135 cm (Stent length mm) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 30 | 40 | 60 | 80 | 100 | 120 | 150 | 170 | 200 | ||
| 6F | 5 | 379898 | 379899 | 379900 | 379901 | 379937 | 379938 | 379939 | 379940 | 379941 |
| 6 | 379903 | 379904 | 379905 | 379906 | 379942 | 379943 | 379944 | 379945 | 379946 | |
| 7 | 379908 | 379909 | 379910 | 379911 | 379947 | 379948 | 379949 | 379950 | 379951 | |
TLR = Target Lesion Revascularization; A.L.L = Average Lesion Length
1.BIOTRONIK data on file. 6.0 mm diameters; 2. BIOTRONIK data on file. 6.0 mm diameters. Supera stent not possible to test due to its design and applied test method; 3. Zhao HQ Late stent expansion and neointimal proliferation of oversized nitinol stents in peripheral arteries. Cardiovasc. Interv. Radiol. 2009; 32(4); 720-6; 4. Koskinas C. Role of endothelial shear stress in stent restenosis and thrombosis: pathophysiologic mechanisms and implications for clinical translation. JACC 2012 10;59(15):1337-49; 5. Koppara T. Thrombogenicity and early vascular healing response in metallic biodegradable polymer-based and fully bioabsorbable drug-eluting stents. Circ Cardiovasc Interv. 2015 8(6):e002427; 6. Funovics M. Correlation between chronic outward force (COF) and neointimal hyperplasia in self-expanding nitinol stents in swine in clinically relevant oversizing ranges. Presented at: LINC, Jan 26, 2017; Leipzig, Germany; 7. Lichtenberg et al. Effectiveness of the Pulsar-18 self-expanding stent with optional drug-coated balloon angioplasty in the treatment of femoropopliteal lesions - the BIOFLEX PEACE All-Comers Registry.Vasa (2019), 1-9. doi_10.10240301-1526a000785; 8.Bosiers M et al. 4-French - compatible endovascular material is safe & effective in the treatment of femoropopliteal occlusive disease: Results of the 4EVER Trial. ENDOVASC THER 2013; 20: 746-756; 9. Lichtenberg M. Superficial Femoral Artery TASC D registry: 12-month effectiveness analysis of the Pulsar-18 SE nitinol stent in patients with critical limb ischemia. J Cardiovasc Surg (Torino). 2013 ; 54(4):433-9; 10. BIOTRONIK data on file.
Leading competitors have been selected based on the PV Stent Revenue Market Shares EU, 2017 and PV Revenue Market Shares APAC 2015; (Source: Millennium Research Group Inc.).
*Indication as per IFU. Pulsar and proBIO are trademarks or registered trademarks of the BIOTRONIK Group of Companies. All other trademarks are the property of their respective owners.