Vascular Intervention //
Coronary
Drug-Coated Balloon Catheter
Pantera® Lux®
Clinically proven. Best in class crossability.

 Clinically proven solution for in-stent restenosis and de novo lesions
Clinically proven solution for in-stent restenosis and de novo lesions
 Lux coating technology for rapid drug absorption
Lux coating technology for rapid drug absorption
 Advanced trackability
Advanced trackability
Clinically proven solution for in-stent restenosis and de novo lesions
Clinical outcomes from multiple studies1-8 including randomized controlled trials like BIOLUX RCT, ISAR-DESIRE 4 and PEBSI show high efficacy and safety for both in-stent restenosis and de novo lesions.
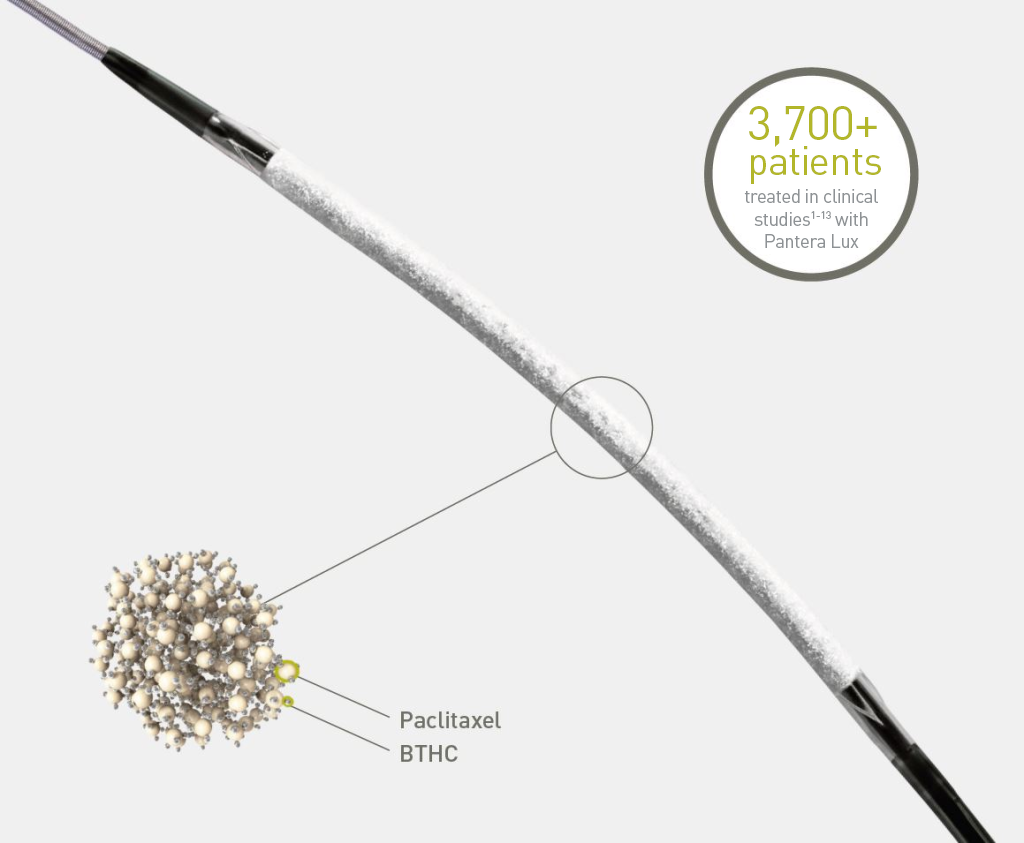
Lux coating technology for rapid drug absorption

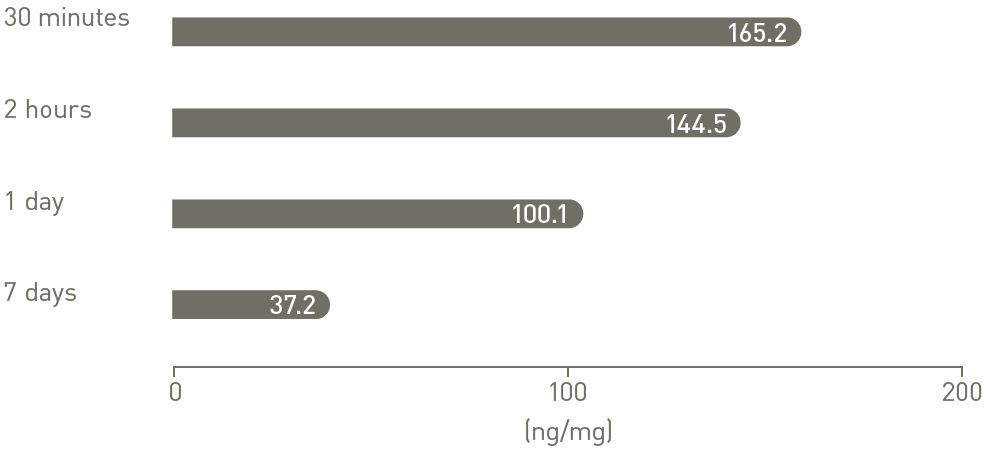
Prolonged tissue retention at the target site9, 10
Pig coronary artery Paclitaxel tissue concentrations
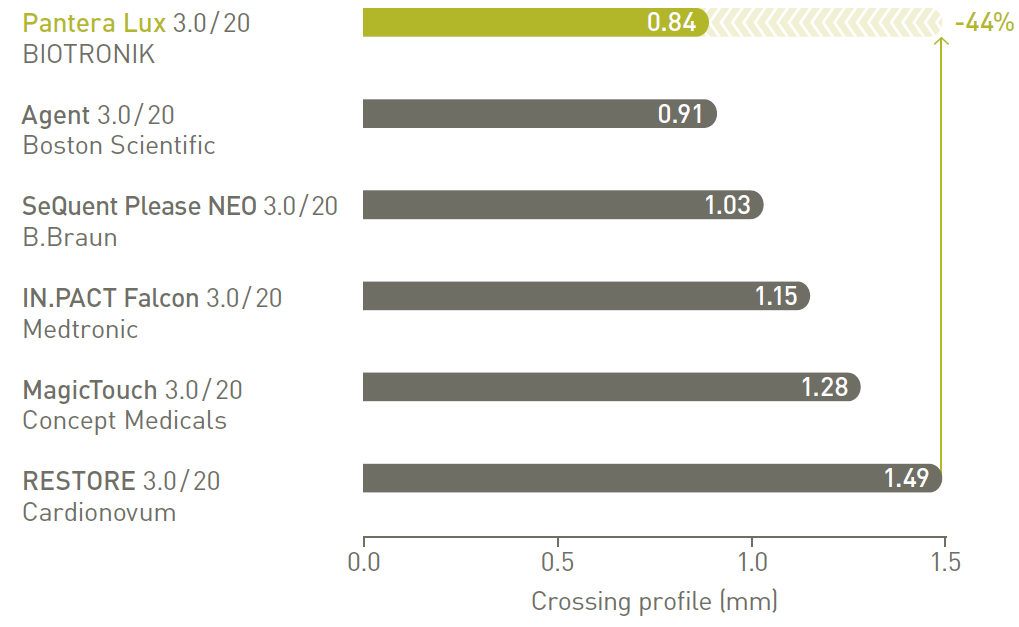
Advanced trackability
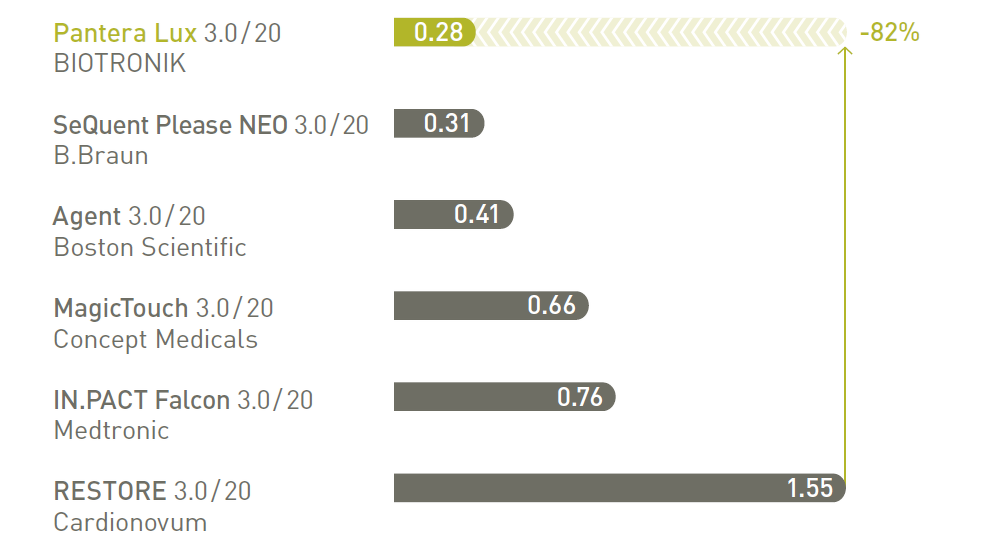
Pantera Lux, with its low crossing profile, provides advanced trackability to successfully reach and treat most cases in daily practice.
Low crossing profile 11
Low track force11
 Pantera Lux
Pantera Lux
Indicated for balloon dilatation for in-stent restenosis, de-novo lesions, acute or impending vascular occlusion and treatment of small vessel disease.*
Technical Data
| Drug-Coated Balloon Catheter | |
|---|---|
| Catheter type |
Fast-exchange PTCA balloon catheter |
| Recommended guide catheter | 5F (min. I.D. 0.056”) |
| Lesion entry profile |
0.017” |
| Guide wire diameter |
0.014” |
| Usable catheter length |
140 cm |
| Balloon folding |
3-fold |
| Ballon markers | Two embedded platinum-iridium markers |
| Brachial shaft marker | 92 cm from tip |
| Femoral shaft marker |
102 cm from tip |
| Proximal shaft diameter |
2.0 F |
| Distal shaft diameter | 2.5 F (ø 2.0 - 3.5 mm), 2.6F (ø 4.0 mm) |
| Nominal Pressure (NP) | 7 atm |
| Rated Burst Pressure (RBP) | 13 atm (ø 2.0 - 3.5 mm); 12 atm (ø 4.0 mm) |
| Coating | |
|---|---|
| Drug | Paclitaxel |
| Drug dose | 3.0 μg/mm² |
| Delivery matrix | Paclitaxel and Butyryl-tri-hexyl citrate (BTHC) |
| Coated area | Cylindrical section of the balloon, exceeding the proximal and distal markers |
Compliance Chart
| Balloon Diameter x Length (mm) | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ø 2.0 x 10-30 |
ø 2.5 x 10-30 |
ø 3.0 × 10-30 |
ø 3.5 × 10-30 |
ø 4.0 × 10-30 |
|||||||||||||||
| Nominal Pressure | atm5 | 7 | 7 | 7 | 7 | 7 | |||||||||||||
| (NP) | ø (mm) | 2.00 | 2.50 | 3.00 | 3.50 | 4.00 | |||||||||||||
| Rated Burst Pressure | atm5 | 13 | 13 | 13 | 13 | 12 | |||||||||||||
| (RBP) | ø (mm) | 2.26 | 2.82 | 3.48 | 4.11 | 4.59 | |||||||||||||
| **1 atm = 1.013 bar | |||||||||||||||||||
Ordering Information
Contact
The Pantera® Lux® DCB with its Lux® coating is part of the Lux® family of Paclitaxel-coated balloons from BIOTRONIK.
1 Hehrlein C. et al. Cardiovasc. Revasc. Med. 2012 Sep; 13(5): 260-4;
2 Toelg R. et al. EuroIntervention 2014 Sep; 10(5): 591-9;
3 Naber C.K. EuroPCR 2016. oral presentation. BIOLUX RCT Clinical performance of the Pantera Lux Paclitaxel coated balloon vs. drug-eluting Orsiro hybrid stent system in patients with in-stent restenosis: a randomized controlled trial;
4 Kufner et al. J Am Coll Cardiol Intv 2017;10: 1332 -40, Clinical trials.gov, NCT01632371;
5 Garcia-Touchard et al. EuroIntervention. 2017 Jan 20;12(13):1587-1594. NCT01839890;
6 Vos N. S. et al. Vos N. S. et al. EuroIntervention 2014;10:584-590;
7 Jim M. H. AsiaPCR 2014, oral presentation, Six-month Angiographic Restudy of Paclitaxel-Eluting balloon kissing in Dealing with side branch Ostial Narrowing (SARPEDON);
8 Worthley S. et al. Cardiovasc. Revasc. Med. 2015; 16: 413-417;
9 Radke P. et al. EuroIntervention. 2011 Oct; 7(6): 730-7;
10 BIOTRONIK data on file;
11 BIOTRONIK data on file, compared to main competitors.
SeQuent is a registered trademark of the B. Braun Group of Companies; ELUTAX is a registered trademark of Aachen Resonance; IN.PACT is a registered trademark of the Medtronic Group of Companies; DIOR is a registered trademark of Eurocor.
*Indication as per IFU (may differ in countries not accepting CE mark).

