EMBRACE LIFE AGAIN PROSPERA® SPINAL CORD STIMULATOR WITH EMBRACE ONE™

THE PAINFUL TRUTH ABOUT CHRONIC PAIN
If you are living with chronic pain, you feel the impact it has on your everyday life. It affects you not just physically, but mentally and emotionally as well. For many, this can mean:
DEPRESSION
ANXIETY
FATIGUE
TROUBLE SLEEPING
UNDERSTANDING SPINAL CORD STIMULATION
As you know, there are many treatment options for chronic pain. One you may be less familiar with is SCS, which works by using small electrical impulses to interrupt pain signals before they reach the brain.
SCS CAN PROVIDE3-5:
- EFFECTIVE PAIN RELIEF
- IMPROVED QUALITY OF LIFE
- IMPROVED OVERALL MOVEMENT
- IMPROVED SLEEP
- REDUCED DEPENDENCE ON MEDICATION
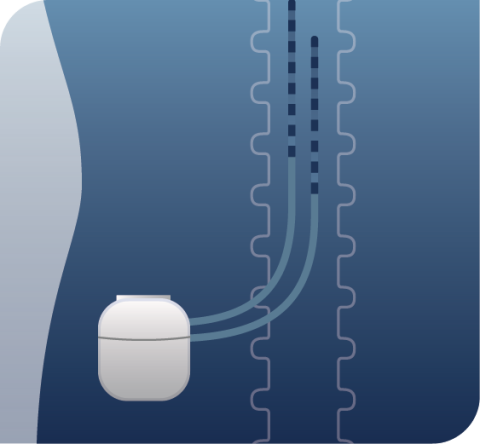
HOW SCS THERAPY WORKS:
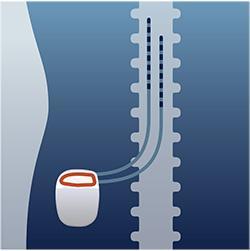
Thin leads are placed near specific nerves in your back and attached to a small implanted stimulator.
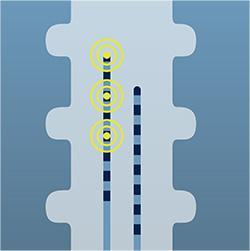
Stimulation travels from the implanted stimulator through the leads to communicate with your nerves.
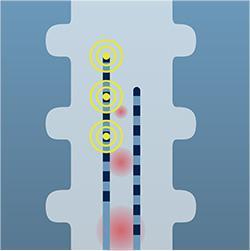
The stimulation is designed to interrupt and mask the pain signals as they travel to the brain.

You are given a programmer, which is a phone-like device that allows you to control your therapy.
THE UNIQUE BIOTRONIK NEURO ADVANTAGE
The Prospera SCS System
Get reliable relief from your chronic pain. Only the Prospera SCS System has RESONANCE™, a proprietary therapy that leverages how the body processes pain. It was designed to stimulate a broader area of your nerves.6
With RESONANCE therapy7,8:
- Patients experienced overall improvements in quality of life, including sleep
- 82% of patients experienced significant overall pain reduction†
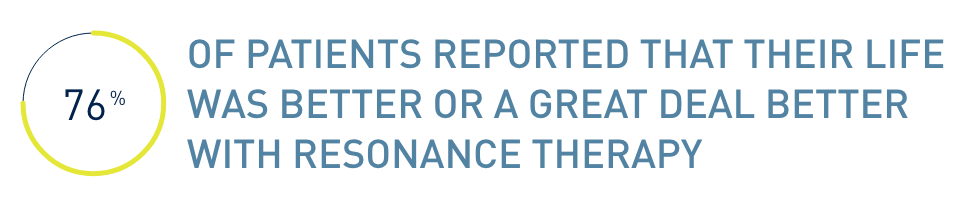
†Defined as ≥50% pain reduction from baseline.

PERSONAL PERSPECTIVES ON PROSPERA
Hear Pain Relief Stories from Real Prospera Patients
April's Story
Bibi's Story
TRY THE PROSPERA SCS SYSTEM TO SEE IF IT’S RIGHT FOR YOU
Unlike other treatment options, you can test SCS therapy during a trial period to see how it works for you before committing long term.
HERE'S HOW THE PROSPERA SCS SYSTEM TRIAL PERIOD WORKS
1. Temporary leads are placed near specific nerves in your back during a simple in-office procedure.

2. The leads are connected to a small, discreet external stimulator you wear under your clothes.

3. You go about your daily activities, which gives you a chance to see how Prospera SCS can easily fit into your life and provide relief.

4. You, your doctor, and your BIOTRONIK Neuro Embrace One Care Team will monitor any pain relief, improvements in function, and other benefits you may experience.

5. If you and your doctor decide Prospera SCS is the right long-term therapy for you, you can rest assured that your dedicated Embrace One Care Team will be with you every step of the way.
MRI for Real Life: Get the Scans You May Need⁹
- FDA-approved SCS system that allows you to get full-body 1.5T and 3T scans in normal operating conditions
- Peace of mind that you have access to the most advanced scans

Proactive Care* with Embrace One™
Embrace One is another unique BIOTRONIK Neuro advantage. It is the only patient support system that allows for automatic, daily, remote monitoring of your therapy. Your Embrace One Care Team is made up of trained, dedicated specialists who monitor your therapy 7 days a week to ensure that it is working the best for you.
95% of patients would choose SCS with daily remote monitoring10
The Embrace One Care Team consists of trained, dedicated specialists who monitor your therapy 7 days a week.
If they detect that any adjustments may be needed, the team will proactively reach out to you.
Your therapy can be fine-tuned for the best possible relief through a remote programming session, anytime and anywhere.
Your doctor and Embrace One Care Team work together to monitor your therapy, ensuring it is working the best for you.
All data is transmitted using state-of-the-art security and protection.
You are supported by the Embrace One Care Team for the lifetime of your therapy.
BioVantage™
BioVantage is an expanded service available only with the Prospera SCS system. If your doctor chooses to participate in BioVantage, you will receive extra support every month to help you experience the consistent pain relief you need to reach your treatment goals. These remote, monthly sessions help keep your doctor closely connected to your care and progress so they can adjust your therapy if needed.
Setting yourself up for Spinal Cord Stimulation Success with BioVantage
Watch this video to learn more about the Prospera Spinal Cord Stimulation System with Embrace One and BioVantage.
BioVantage offers you:
Regular monthly, in-depth check-ins with your Embrace One care team.
You can use this time to talk about how your SCS therapy is going, whether you’re getting the pain relief you need, how you are progressing toward your treatment goals, and if any changes may be needed to your device.
Sessions scheduled at your convenience and from the comfort of your home.
No need to make a doctor’s appointment, arrange transportation, or take time off from work. Monthly calls with the Embrace One care team will be scheduled at a convenient time, and you can easily join from wherever you are. Each session could include a copay similar to what you would expect for an office visit.
Doctor-recommended care.
Pain experts agree that these regular check-ins provide an extra level of care that may help improve your quality of life.11 Monthly check-ins also allow your doctor to fine-tune your therapy based on how you’re doing.
Embrace One is a support platform intended to help manage a patient’s experience with spinal cord stimulation. It is not intended to be used for medical diagnosis or medical treatment.
*Proactive Care: BIOTRONIK Neuro’s remote support team may reach out to patients to ensure proper usage of the spinal cord stimulator based on remotely monitored data. BIOTRONIK Neuro does not provide health advice or clinical actions outside the scope of spinal cord stimulator proper usage. This product support is not a replacement for the patient’s responsibility to communicate any medical questions or concerns with the physician’s office.
BioVantage is an elective, paid subscription offering to healthcare providers from BIOTRONIK Neuro, Inc. All services are provided under the general supervision of a physician. Patients should consult with their healthcare provider about BioVantage enrollment.
REFERENCES: 1. Institute of Medicine (US) Committee on Advancing Pain Research, Care, and Education. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington (DC): National Academies Press (US); 2011. 2. International Neuromodulation Society. Accessed March 16, 2023. https://www.neuromodulation.com/spinal-cord-stimulation 3. Grider JS, Manchikanti L, Carayannopoulos A, et al. Effectiveness of spinal cord stimulation in chronic spinal pain: a systematic review. Pain Physician. 2016;19(1):E33-E54. 4. Pollard EM, Lamer TJ, Moeschler SM, et al. The effect of spinal cord stimulation on pain medication reduction in intractable spine and limb pain: a systematic review of randomized controlled trials and meta-analysis. J Pain Res. 2019;12:1311-1324. 5. Manca A, Kumar K, Taylor RS, et al. Quality of life, resource consumption and costs of spinal cord stimulation versus conventional medical management in neuropathic pain patients with failed back surgery syndrome (PROCESS trial). Eur J Pain. 2008;12(8):1047-1058. 6. Lim K, et al. Functional ultrasound imaging reveals activation properties of clinical spinal cord stimulation therapy stimulation programming. J Pain Res. In press. 7. Russo M, et al. Long-term study of SCS system with multiphase stimulation and remote device management: interim 24-month results. North American Neuromodulation Society Annual Meeting; January 30-February 1, 2025; Orlando, FL. 8. Data on file. BIOTRONIK Neuro. 9. Prospera Spinal Cord Stimulation System, MRI Guidelines Technical Manual. Document 461825, Revision C. Biotronik Neuro. Lake Oswego, OR; July 2022. 10. Russo M, et al. Remote SCS management reduces patient burdens and improves care: interim 24-month study results. North American Neuromodulation Society Annual Meeting; January 30-February 1, 2025; Orlando, FL. 11. Staats P, Deer TR, et al. Remote management of spinal cord stimulation devices for chronic pain: expert recommendations on best practices for proper utilization and future considerations. Neuromodulation. 2023;26:1295-1308.
Brief Summary: Please reference the appropriate product Instructions for Use (IFU) for more information regarding indications, contraindications, warnings, precautions, and potential adverse events. Indications for Use: The Prospera® Spinal Cord Stimulation (SCS) System is indicated as an aid in the management of chronic, intractable pain in the trunk and/or limbs, which may include unilateral or bilateral pain. Contraindications: Implantation of a spinal cord stimulator may be contraindicated in patients who are unable to operate the SCS system, or who have failed to receive effective pain relief during SCS trial stimulation, or who are poor candidates for surgery. Note that the safety and effectiveness of Prospera SCS system has not been established in pediatric patients or pregnant or nursing patients. Warnings: The following may cause electromagnetic interference, adverse interactions, insufficient or excessive stimulation, damage and function loss of the system, and/or therapy failure: external defibrillation, transcutaneous electrical nerve stimulation (TENs), lithotripsy, RF ablation, hyperbaric oxygen therapy, electrocautery, diathermy therapy (including shortwave, microwave, and therapeutic ultrasound therapies), high-power ultrasound, radiation therapy, Magnetic Resonance Imaging (MRI) scan (refer to Prospera SCS System MRI Guidelines for the system’s MR conditional information), use of portable RF communication equipment near the SCS system, use of a non-BIOTRONIK-provided charger. The Prospera SCS System may interfere with the operation of implanted pacemakers or ICDs. The effects of an implanted Prospera SCS System on other neurostimulators are unknown. Precautions: Device malfunction, loss of therapy, and other adverse events including patient injury may occur if the device is not handled or operated properly as described in the IFU. Refer to the product IFU for comprehensive safety messages when handling the device. Potential Adverse Events: Risks associated with SCS system placement: pain at the implant site, infection, cerebrospinal fluid (CSF) leakage, CSF fistula, epidural hemorrhage, bacterial meningitis, seroma, hematoma, paralysis. Additional risks associated with SCS system use: lead migration; stimulator migration; allergic response or tissue reaction to the implanted system material; skin erosion; radicular chest wall stimulation; disturbed urination; dysesthesia; decubitus; premature battery depletion; and uncomfortable stimulation or ineffective pain management. Furthermore, there is the risk that the SCS therapy may not be effective in relieving symptoms or may cause worsening of symptoms. Refer to the product IFU for a comprehensive list of potential adverse events.
