
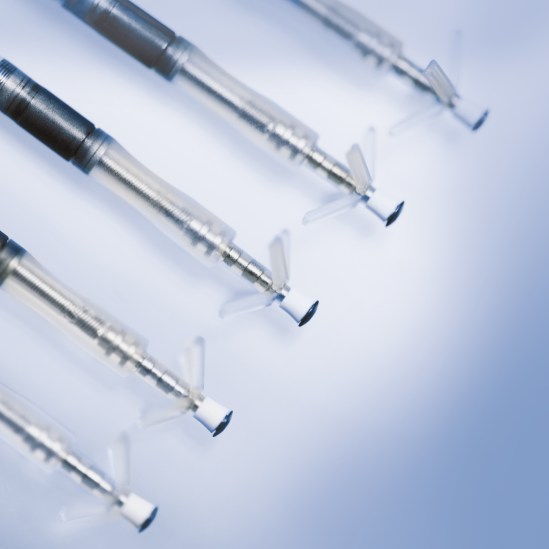

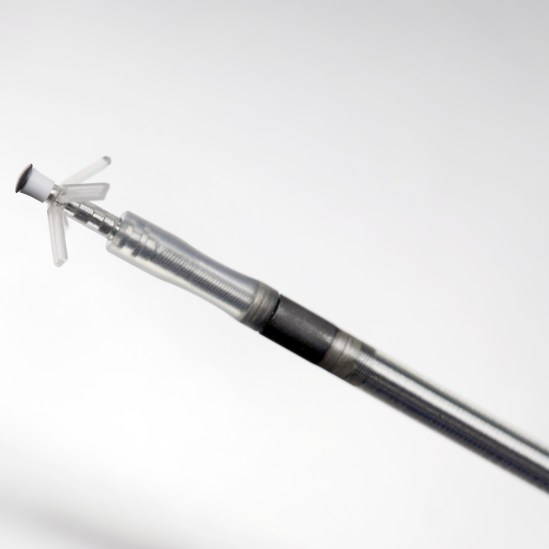






Solia ProMRI
Active and Passive Fixation MR Conditional Pacing Leads
BIOTRONIK continues to expand its ProMRI portfolio by introducing two passive fixation pacing leads into the US market. Solia T and Solia JT provide patients the assurance that they can get the device therapy they need, as well as access to future diagnostics, to support their best overall health conditions.
The Solia family of pacing leads offers 5.6 F lead body diameter (6 F introducer) that is MR conditional, together with a polyurethane sleeve over a silicon insulator designed for reduced friction. Solia leads are approved for use with all BIOTRONIK ProMRI systems, giving physicians more options to accommodate varying patient anatomies.
Solia leads received FDA approval for use based on the SIELLO Clinical Study, in which 1,758 patients were enrolled at 60 US sites, with 3,220 implanted leads. To date, over 1 million Solia/Siello leads have been implanted worldwide. BIOTRONIK’s lead reliability record exemplifies commitment to quality, including no lead recalls, ever.
