Orsiro®Mission
Orsiro Mission DES is indicated for improving coronary luminal diameter in patients with symptomatic ischemic heart disease due to discrete de-novo stenotic lesions and in-stent restenotic lesions (length ≤ 40 mm) in the native coronary arteries with a reference vessel diameter of 2.25 mm to 4.0 mm including the following patient and lesion subsets:
- Acute Coronary Syndrome (ACS)
- ST-Elevation Myocardial Infarction (STEMI)
- Diabetes Mellitus (DM)
- Complex Lesions (B2/C)
- High Bleeding Risk (HBR)
- Long Lesions (LL) (e.g. ≥ 20 mm)
- Small Vessels (SV) (e.g. ≤ 2.75 mm)
- Multi-Vessel Disease (MVD)
- Male/Female
- Old Patients (e.g. > 65 y)
Product Highlights
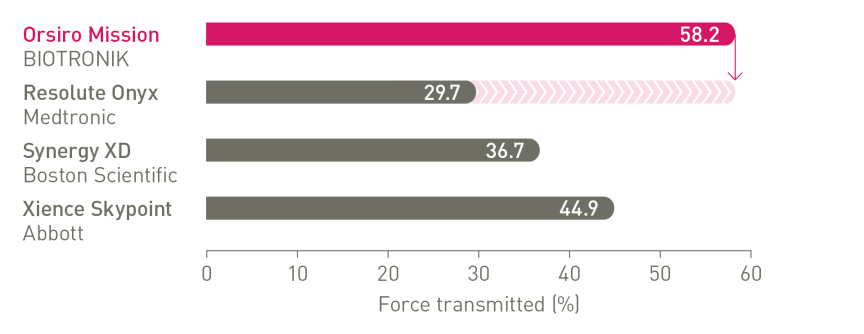
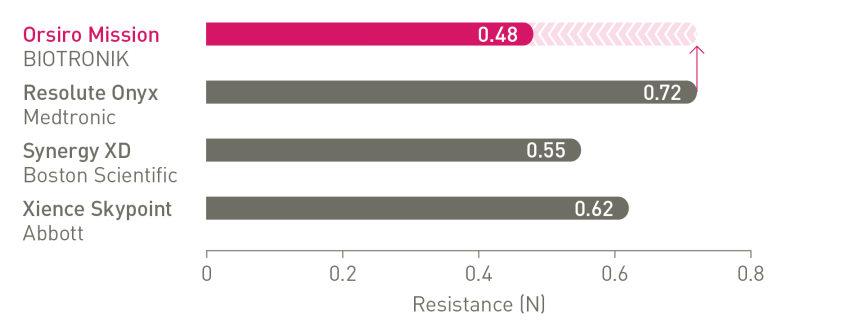
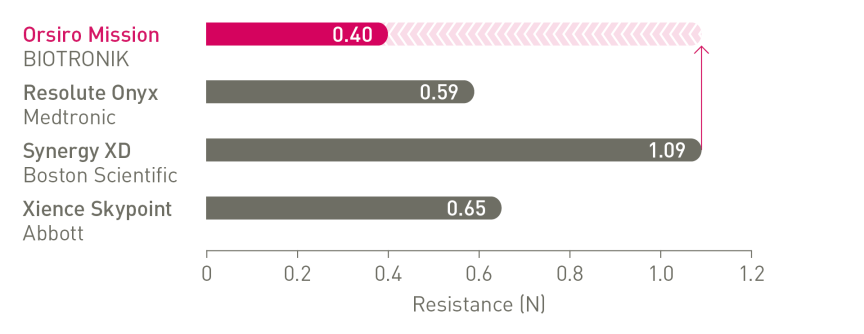
The next level of deliverability¹
1st in Push, Track and Cross4
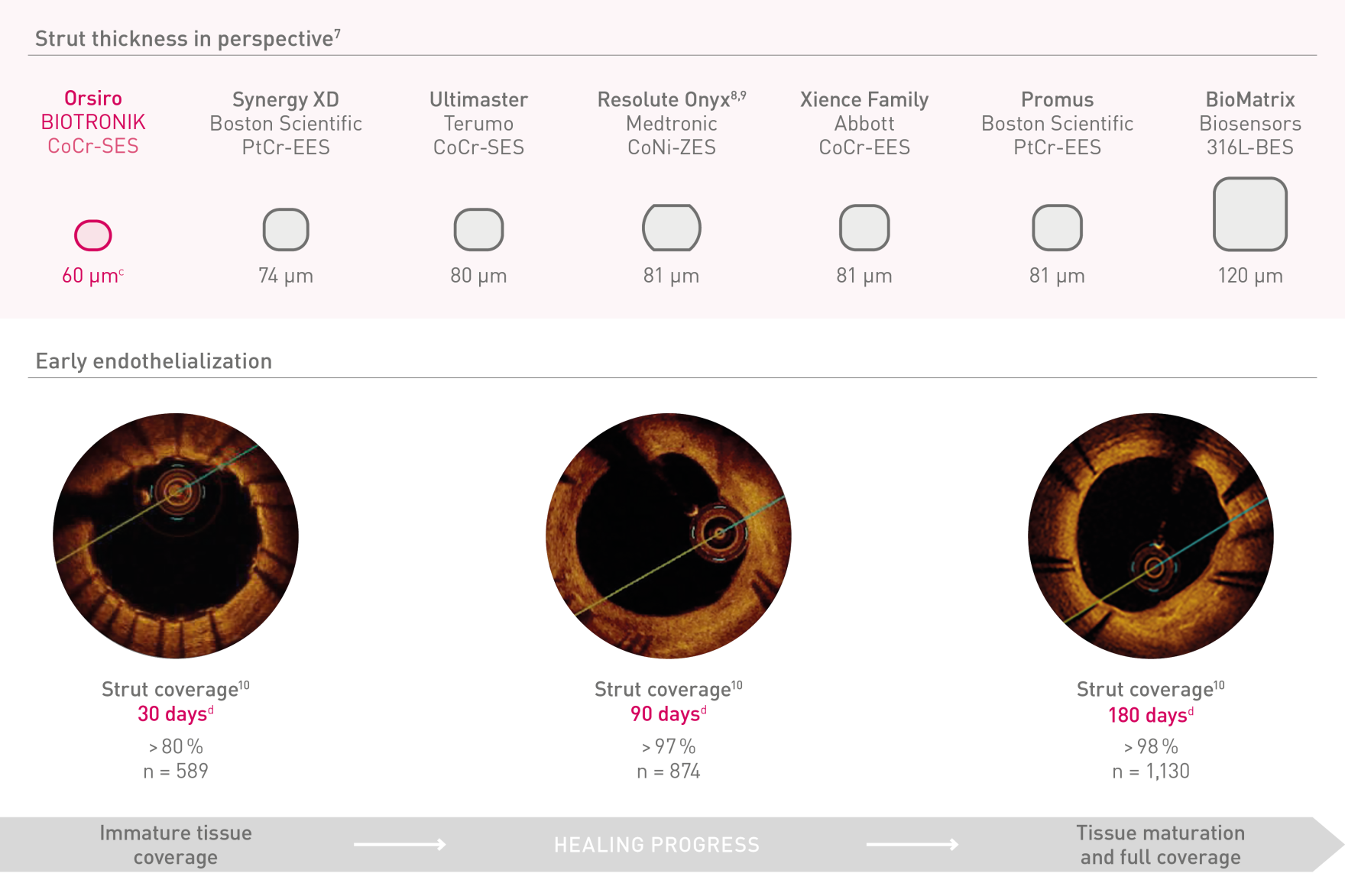
Ultrathin struts²
For early endothelialization5
Outstanding patient outcomes³
Proven Superiority in STEMI6
Continued Superiority in STEMI at 2 yearsf6
In the BIOSTEMI trial, Orsiro is superior to Xience in STEMI patients with respect to the primary endpoint of Target Lesion Failure (TLF) at 24 months.
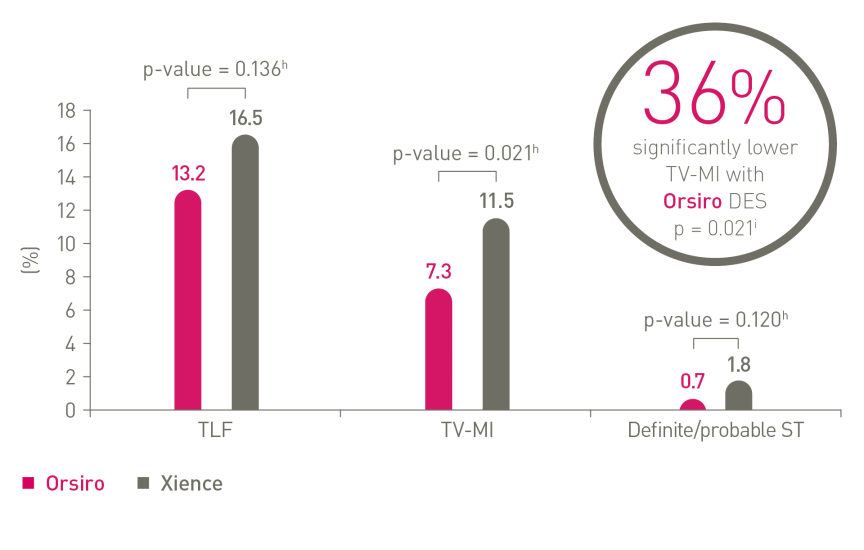
Pushing the boundaries of safety performance with Orsirog
In the 5-Year results of the FDA pivotal trial, BIOFLOW-V, Orsiro ultrathin-strut DES showed significantly less target vessel myocardial infarction events compared to Xience DP-EES.12
Extensive Clinical Program of the Orsiro DES Familyj13
Product Overview
Orsiro Mission
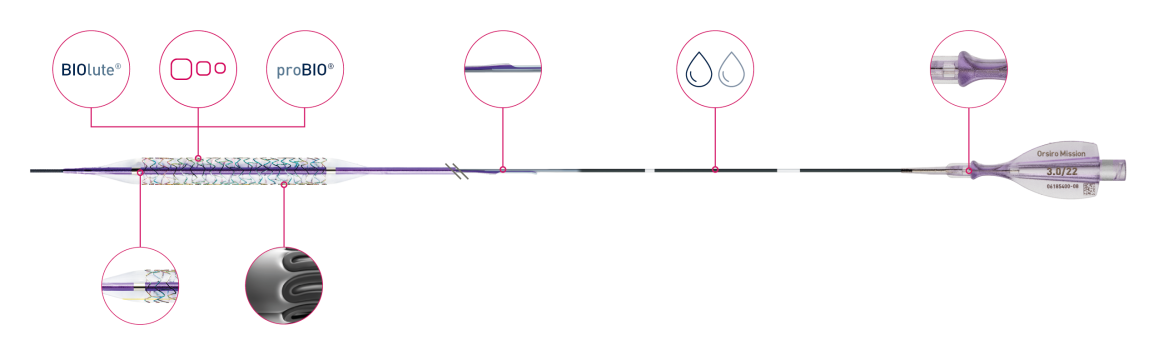
Bioabsorbable coating
Ultrathin 60 µmᶜ struts
Passive coating
Enhanced force transmission
Dual-coating
Ergonomic hub
Flexible shaft
Deep embedding
Technical Data
| Stent | |
|---|---|
| Stent material | Cobalt chromium, L-605 |
| Strut thickness | ø 2.25 – 3.0 mm: 60 µm (0.0024”); ø 3.50 – 4.0 mm: 80 µm (0.0031”) |
| Passive coating | proBIO® (Amorphous Silicon Carbide) |
| Active coating | BIOlute® bioabsorbable Poly-L-Lactide (PLLA) eluting a limus drug |
| Drug dose | 1.4 µg/mm² |
| Delivery system | |
| Catheter type | Rapid exchange |
| Recommended guide catheter | 5F (min. I.D. 0.056”) |
| Guide wire diameter | 0.014” |
| Usable catheter length | 140 cm |
| Balloon material | Semi crystalline polymer material |
| Coating (Distal shaft) | Hydrophilic |
| Coating (Proximal shaft) | Hydrophobic |
| Marker bands | Two swaged platinum-iridium markers |
| Lesion entry profile | 0.017” |
| Distal shaft diameter | 2.7F: ø 2.25 – 3.0 mm; 2.9F: ø 3.5 – 4.0 mm |
| Proximal shaft diameter | 2.0F |
| Nominal pressure (NP) | 10 atm |
| Rated burst pressure (RBP) | 16 atm |
| Storage | |
|---|---|
| Use Before Date (UBD) | 24 months |
| Temperature | Between 15°C (59°F) and 25°C (77°F), short term excursions between 10°C (50°F) and 40°C (104°F) are allowed |
Ordering Information
| Stent ø (mm) |
Stent Length (mm) |
||||||||
|---|---|---|---|---|---|---|---|---|---|
| 9 | 13 | 15 | 18 | 22 | 26 | 30 | 35 | 40 | |
| 2.25 | 419101 | 419107 | 419113 | 419119 | 419125 | 419131 | 419137 | 419143 | 419149 |
| 2.5 | 419102 | 419108 | 419114 | 419120 | 419126 | 419132 | 419138 | 419144 | 419150 |
| 2.75 | 419103 | 419109 | 419115 | 419121 | 419127 | 419133 | 419139 | 419145 | 419151 |
| 3.0 | 419104 | 419110 | 419116 | 419122 | 419128 | 419134 | 419140 | 419146 | 419152 |
| 3.5 | 419105 | 419111 | 419117 | 419123 | 419129 | 419135 | 419141 | 419147 | 419153 |
| 4.0 | 419106 | 419112 | 419118 | 419124 | 419130 | 419136 | 419142 | 419148 | 419154 |
Downloads and Related Links
Downloads
Related Links
Media
Product Animations
How can we help you?
References
a. In comparison to Resolute Onyx, BIOTRONIK data on file; b. In comparison to Synergy XD, BIOTRONIK data on file; c. Ø2.25-3.0mm; d. Images: Secco G et al. Time-related changes in neointimal tissue coverage following a new generation SES implantation: an OCT observational study. Presented at: euro PCR, May 20, 2014; Paris, France; e. Bayesian Credible Interval; f. In comparison to Xience, based on TLF, in the BIOSTEMI trial at 2 years; g. In comparison to Xience, based on statistically significant lower TV-MI and late/very late definite/probable ST rates from the BIOFLOW-V trial through 5 years; h. p-values for 60-month frequentist analysis; i In comparison to Xience, based on BIOFLOW-V 5-year results; j. Orsiro and Orsiro Mission DES; k. All-comers patients, BIO-RESORT 5 year outcomes; l. Based on 1-year TLF SUCRA (Surface Under the Cumulative Ranking Curve), Taglieri Network Meta-Analysis; m. In comparison to Xience, based on a Rate Ratio of 0.58, in the BIOSTEMI trial at 2 years.
1. In comparison to Xience Sierra, Resolute Onyx and Synergy for bench tests on pushability, trackability and crossability, BIOTRONIK data on file; 2. As characterized with respect to strut thickness in Bangalore et al. Meta-analysis; 3. Based on investigator’s interpretation of BIOFLOW-V primary endpoint result; 4. BIOTRONIK data on file; 5. Per investigators’ interpretation in Secco et al. Imaging data serial observations. Secco GG et al. Time-related changes in neointimal tissue coverage of a novel Sirolimus eluting stent: Serial observations with optical coherence tomography. Cardiovascular Revascularization Medicine. 2016; 17(1): 38-43; 6. Pilgrim et al. Biodegradable – versus durable-polymer drug-eluting stents for STEMI. Final 2-year outcomes of the BIOSTEMI trial. J Am Coll Cardiol. Cardiovasc Interven. 2021, doi: 10.1016/j.jcin.2020.12.011; 7. Stefanini GG et al. Coronary stents: novel developments. Heart. 2014 Jul 1;100(13):1051-61; 8. Low AF. Stent platform for procedural success: Introducing the Continuous Sinusoidal & Core Wire Technologies. Presented at: AsiaPCR; 22-24 January, 2015; Singapore, Singapore; 9. Tolentino A. Evolving DES Strategy: Biodegradable Polymer vs. Bioabsorbable Scaffold. Presented at: Cardiovascular Nurse/Technologist Symposium; June 17, 2016; New York, USA; 10. Secco G et al. Time-related changes in neointimal tissue coverage of a novel Sirolimus eluting stent: Serial observations with optical coherence tomography. Cardiovascular Revascularization Medicine 17.1 (2016): 38-43; 11. Iglesias JF, et al. Biodegradable polymer sirolimus-eluting stents versus durable polymer everolimus-eluting stents in patients with ST-segment elevation myocardial infarction (BIOSTEMI) The Lancet. 2019 Oct 5;394(10205):1243-53; 12. Kandzari D et al. Ultrathin Bioresorbable Polymer Sirolimus-Eluting Stents versus Thin Durable Polymer Everolimus-Eluting Stents for Coronary Revascularization: Final 5-year Outcomes from the Randomized BIOFLOW V Trial, Submitted manuscript to JACC, 2022: NCT02389946; 13. BIOTRONIK data on file, status February 2023; 14. Per investigators’ interpretation of preclinical studies with Orsiro as mentioned in Cassese et al. J Thorac Dis 2018;10(2):688-692; 15. E.Ploumen, BIO-RESORT 5 year outcomes and diabetic subgroup, TCT 2021; 16. Taglieri N et al. Target lesion failure with current drug-eluting stents: Evidence from a comprehensive network meta-analysis. JACC 2020 13(24):2868-78.
Clinical data collected with the Orsiro DES device within the Orsiro family clinical program
Orsiro, Orsiro Mission, proBIO and BIOlute are trademarks or registered trademarks of the BIOTRONIK Group of Companies. All other trademarks are the property of their respective owners.