Highly pushable coaxial shaft design
A strong inner shaft and a flexible outer shaft creating a highly pushable and deliverable system.
Up to 9% higher pushability compared to leading competitors.1
Passeo-18 is indicated to dilate stenosis in the femoral, popliteal and infrapopliteal arteries and for the treatment of obstructive lesions of native or synthetic arteriovenous dialysis fistulae.*
Coaxial shaft design with up to 9% higher pushability compared to leading competitors.1
Predictable radial balloon expansion to minimize the risk of dissection.
Wide balloon diameters up to ø7.0mm and long balloon lengths up to 200mm.
A strong inner shaft and a flexible outer shaft creating a highly pushable and deliverable system.

Up to 9% higher pushability compared to leading competitors.1
Low, controlled compliance for predictable radial balloon expansion to minimize the risk of dissection.
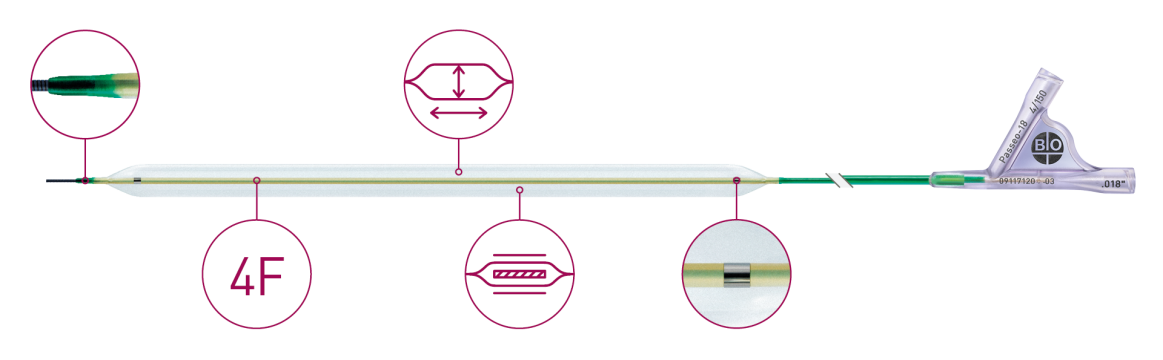
The available low profile 3.8F coaxial catheter shaft design facilitates access to distal lesions and allows reduction of access site complications.2
| Balloon catheter | |
|---|---|
| Catheter type | OTW |
| Recommended guide wire | 0.018” |
| Tip | Short and tapered, colored |
| Balloon material | SCP (Semi-Crystalline Polymer), controlled compliance (4 - 8 %) |
| Balloon folding | 5-fold |
| Balloon coating | Hydrophobic patchwork coating |
| Balloon markers | 2 swaged markers (zero profile) |
| Sizes | ø 2.0 - 7.0 mm; L: 20 - 200 mm |
| Shaft | 3.8F, 3.9F (ø 6.0 /7.0 mm x 170 - 200 mm); coaxial design |
| Usable length | 90, 130 and 150 cm |
| Balloon diameter x length (mm) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| ø 2.0 x 20-170 |
ø 2.0 x 200 |
ø 2.5 x 20-170 |
ø 2.5 x 200 |
ø 3.0 x 20-170 |
ø 3.0 x 200 |
ø 3.5 x 20-170 |
ø 3.5 x 200 |
||
| Nominal Pressure (NP) |
atm* ø (mm) |
6 2.0 |
6 2.0 |
6 2.5 |
6 2.5 |
6 3.0 |
6 3.0 |
6 3.5 |
6 3.5 |
| Rated Burst Pressure (RBP) |
atm* ø (mm) |
15 2.1 |
14 2.1 |
15 2.6 |
14 2.6 |
15 3.2 |
14 3.2 |
15 3.7 |
14 3.7 |
| Balloon diameter x length (mm) | |||||||||
| ø 4.0 x 20-150 |
ø 4.0 x 170-200 |
ø 5.0 x 20-120 |
ø 5.0 x 150 |
ø 5.0 x 170-200 |
ø 6.0 x 20-200 |
ø 7.0 x 20-200 |
|||
| Nominal Pressure (NP) |
atm* ø (mm) |
6 4.0 |
6 4.0 |
6 5.0 |
6 5.0 |
6 5.0 |
6 6.0 |
6 7.0 |
|
| Rated Burst Pressure (RBP) |
atm* ø (mm) |
15 4.3 |
13 4.2 |
15 5.3 |
12 5.2 |
13 5.2 |
12 6.2 |
12 7.3 |
|
| *1 atm = 1.013 bar | |||||||||
| Catheter length (cm) | Balloon ø (mm) | Balloon Length (mm) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 40 | 60 | 80 | 120 | 150 | 170 | 200 | |||
| 4F | 90 | 2.0 | 366098 | 366099 | 366100 | 366104 | 366105 | 366106 | 366114 | 376276 |
| 90 | 2.5 | 357451 | 357458 | 366101 | 357469 | 357476 | 366107 | 357483 | 376277 | |
| 90 | 3.0 | 357452 | 357459 | 366102 | 357470 | 357477 | 366108 | 357484 | 376278 | |
| 90 | 3.5 | 357453 | 357460 | 366103 | 357471 | 357478 | 366109 | 357485 | 376279 | |
| 5F | 90 | 4.0 | 357454 | 357461 | 357465 | 357472 | 357479 | 366110 | 376272 | 376280 |
| 90 | 5.0 | 357455 | 357462 | 357466 | 357473 | 357480 | 366111 | 376273 | 376281 | |
| 90 | 6.0 | 357456 | 357463 | 357467 | 357474 | 357481 | 366112 | 376274 | 376282 | |
| 90 | 7.0 | 357457 | 357464 | 357468 | 357475 | 357482 | 366113a | 376275a | 376283a | |
| Catheter length (cm) | Balloon ø (mm) | Balloon Length (mm) | ||||||||
| 4F | 150 | 2.0 | 366115 | 366118 | 366119 | 366123 | 366126 | 366129 | 366137 | 376296 |
| 130 | 2.5 | 357486 | 357491 | 366120 | 357502 | 357507 | 366130 | 357512 | 376297 | |
| 130 | 3.0 | 357487 | 357492 | 366121 | 357503 | 357508 | 366131 | 357513 | 376298 | |
| 130 | 3.5 | 357488 | 357493 | 366122 | 357504 | 357509 | 366132 | 357514 | 376299 | |
| 5F | 130 | 4.0 | 357489 | 357494 | 357498 | 357505 | 357510 | 366133 | 376292 | 376300 |
| 130 | 5.0 | 357490 | 357495 | 357499 | 357506 | 357511 | 366134 | 376293 | 376301 | |
| 130 | 6.0 | 366116 | 357496 | 357500 | 366124 | 366127 | 366135 | 376294 | 376302 | |
| 130 | 7.0 | 366117 | 357497 | 357501 | 366125 | 366128 | 366136a | 376295a | 376303a | |
| a8 weeks pre-order only | ||||||||||
*Indication as per IFU.
1. BIOTRONIK data on file. Compared to leading competitors 3.0 mm diameter balloon size; 2. Bosiers M, Deloose K, Callaert J, et al. 4-French-compatible endovascular material is safe and effective in the treatment of femoropopliteal occlusive disease: results of the 4-EVER trial. J Endovasc Ther. 2013; 20: 746-756.
Passeo is a trademark or registered trademark of the BIOTRONIK Group of Companies. All other trademarks are the property of their respective owners.