Lower entry profile and advanced tip profile to facilitate crossing
Up to 0.16 mm lower tip entry profile compared to leading competitors2
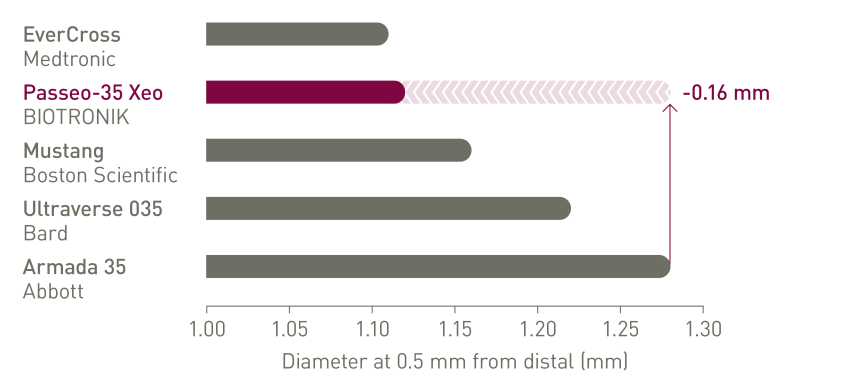
Lowest crossing profile
Up to 0.22 mm lower crossing profile compared to leading competitors2
With usable lengths up to 170cm, Passeo-35 Xeo balloon allows a radial approach1 to dilate stenosis in the iliac*, femoral, popliteal and infrapopliteal arteries. Passeo-35 Xeo balloon is also recommended for post-dilation of stents in the peripheral vasculature and for the treatment of obstructive lesions of native or synthetic arteriovenous dialysis fistulae.**
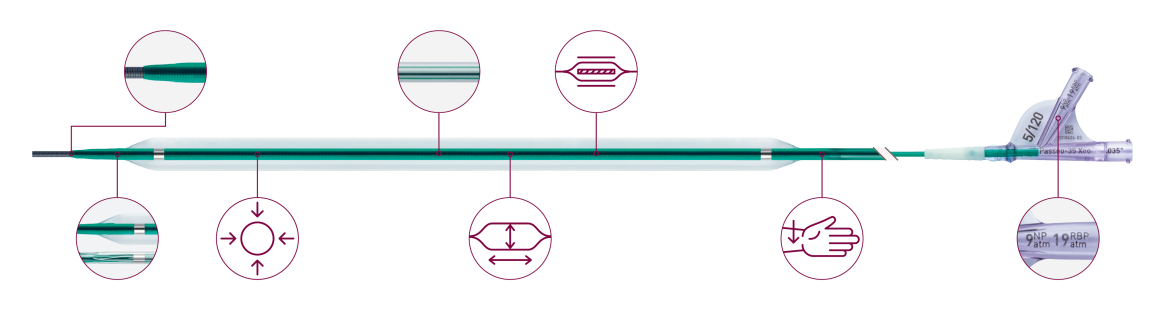
Advanced tip design and low crossing profile
Great trackability and strong pushability
Full 5F compatibility up to 7.0 x 250 mm
Up to 0.16 mm lower tip entry profile compared to leading competitors2

Up to 0.22 mm lower crossing profile compared to leading competitors2
Up to +19.5% more pushability when compared to competitive 0.035’’ PTA balloon catheters3
Low profile design with full 5F compatibility up to 7.0 mm balloon diameter and 250 mm balloon length, 6F up to 10.0 mm and 7F up to 12.0 mm diameter.
| Balloon Catheter | |
|---|---|
| Catheter type | OTW |
| Recommended guide wire | 0.035'' |
| Tip | Low entry profile, colored |
| Balloon material | SCP (Semi-Crystalline Polymer), controlled compliance |
| Balloon coating |
Hydrophobic patchwork coating |
| Balloon markers | 2 swaged markers |
| Sizes | ø 3.0-12.0 mm; L: 20-250 mm |
| Shaft | 5.1-5.4F, dual-lumen, hydrophobic coating |
| Usable length | 90, 130 and 170 cm |
| Guide wire lumen | Hydrophobic coating |
| Balloon ø (mm) | NP¤ | RBP¤ | Catheter length 90 cm Balloon length (mm) |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 40 | 60 | 80 | 100 | 120 | 150 | 170 | 200 | 250 | ||||
| 5F | 3 | 9 | 21 | 428777 | 428786 | 428795 | 428804 | - | - | - | - | - | - |
| 4 | 9 | 20 | 428778 | 428787 | 428796 | 428805 | 428814 | 428823 | - | - | - | - | |
| 5 | 9 | 19 | 428779 | 428788 | 428797 | 428806 | 428815 | 428824 | 428833 | - | 428843 | 428848 | |
| 6 | 9 | 16 | 428780 | 428789 | 428798 | 428807 | 428816 | 428825 | 428834 | - | 428844 | 428849 | |
| 7 | 9 | 15 | 428781 | 428790 | 428799 | 428808 | 428817 | - | - | - | - | - | |
| 6F | 8 | 9 | 14 | 428782 | 428791 | 428800 | 428809 | 428818 | - | - | - | - | - |
| 9 | 9 | 12 | 428783 | 428792 | 428801 | 428810 | 428819 | - | - | ||||
| 10 | 8 | 11 | 428784 | 428793 | 428802 | 428811 | 428820 | - | - | - | - | - | |
| 7F | 12 | 8 | 10 | - | 428794 | 428803 | 428812 | 428821 | - | - | - | - | - |
| Balloon ø (mm) | NP¤ | RBP¤ | Catheter length 130 cm Balloon length (mm) |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 40 | 60 | 80 | 100 | 120 | 150 | 170 | 200 | 250 | ||||
| 5F | 3 | 9 | 21 | 428851 | 428860 | 428869 | 428878 | 428887 | 428896 | 428905 | 428910 | 428915 | 428920 |
| 4 | 9 | 20 | 428852 | 428861 | 428870 | 428879 | 428888 | 428897 | 428906 | 428911 | 428916 | 428921 | |
| 5 | 9 | 19 | 428853 | 428862 | 428871 | 428880 | 428889 | 428898 | 428907 | 428912 | 428917 | 428922 | |
| 6 | 9 | 16 | 428854 | 428863 | 428872 | 428881 | 428890 | 428899 | 428908 | 428913 | 428918 | 428923 | |
| 7 | 9 | 15 | 428855 | 428864 | 428873 | 428882 | 428891 | 428900 | 428909 | 428914 | 428919 | 428924 | |
| 6F | 8 | 9 | 14 | 428856 | 428865 | 428874 | 428883 | 428892 | 428901 | - | - | - | - |
| 9 | 9 | 12 | 428857 | 428866 | 428875 | 428884 | 428893 | 428902b | |||||
| 10 | 8 | 11 | 428858 | 428867 | 428876 | 428885 | 428894 | 428903b | - | - | - | - | |
| 7F | 12 | 8 | 10 | - | 428868 | 428877 | 428886 | 428895 | 428904b | - | - | - | - |
| Balloon ø (mm) | NP¤ | RBP¤ | Catheter length 170 cm Balloon length (mm) |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 40 | 60 | 80 | 100 | 120 | 150 | 170 | 200 | 250 | ||||
| 5F | 4 | 9 | 20 | - | 428935b | 428944b | 428953b | - | 428971b | 428980b | - | 428990b | 428995b |
| 5 | 9 | 19 | 428927b | 428936b | 428945b | 428954b | - | 428972b | 428981b | - | 428991b | 428996b | |
| 6 | 9 | 16 | 428928b | 428937b | 428946b | 428955b | - | 428973b | 428982b | - | 428992b | 428997b | |
| 7 | 9 | 15 | 428929b | 428938b | 428947b | 428956b | - | - | - | - | - | - | |
| 6F | 8 | 9 | 14 | 428930b | 428939b | 428948b | 428957b | - | - | - | - | - | - |
| 9 | 9 | 12 | - | 428940b | 428949b | 428958b | - | - | - | - | - | - | |
| 10 | 8 | 11 | - | 428941b | 428950b | 428959b | - | - | - | - | - | - | |
| ¤NP = Nominal Pressure and RBP = Rated Burst Pressure in atm (1 atm = 1.013 bar) b8 weeks pre-order only |
|||||||||||||
*Note for Australia: Passeo-35 Xeo not approved by Therapeutic Goods Administration for use in the common iliac arteries. **Indication as per IFU.
1. Truesdell et al. Interv Cardiol 2015;7(1)55-76.pdf; 2. BIOTRONIK data on file. Compared to leading competitors 6.0 mm diameter balloon size. IIB(P) 39/2017; 3. BIOTRONIK data on file. Compared to leading competitors 6.0x200 mm balloon size.
Passeo and Xeo are trademarks or registered trademarks of the BIOTRONIK Group of Companies. All other trademarks are the property of their respective owners.