BIOMAG-I Two-Year Study Results Confirm Excellent Safety and Efficacy Profile for Freesolve, BIOTRONIK’s Newest Resorbable Magnesium Scaffold Innovation New Clinical Outcomes Show Continuous Favorable Low Target Lesion Failure and Target Lesion Revascularization
New two-year follow-up data from the BIOMAG-I first-in-human trial confirms a reliable and predictable long-term safety profile for Freesolve™, the third-generation resorbable magnesium scaffold (RMS), establishing it as a true alternative to contemporary drug-eluting stents (DES). The results were presented by Prof. Dr. Michael Haude at the EuroPCR 2024 conference yesterday in Paris.
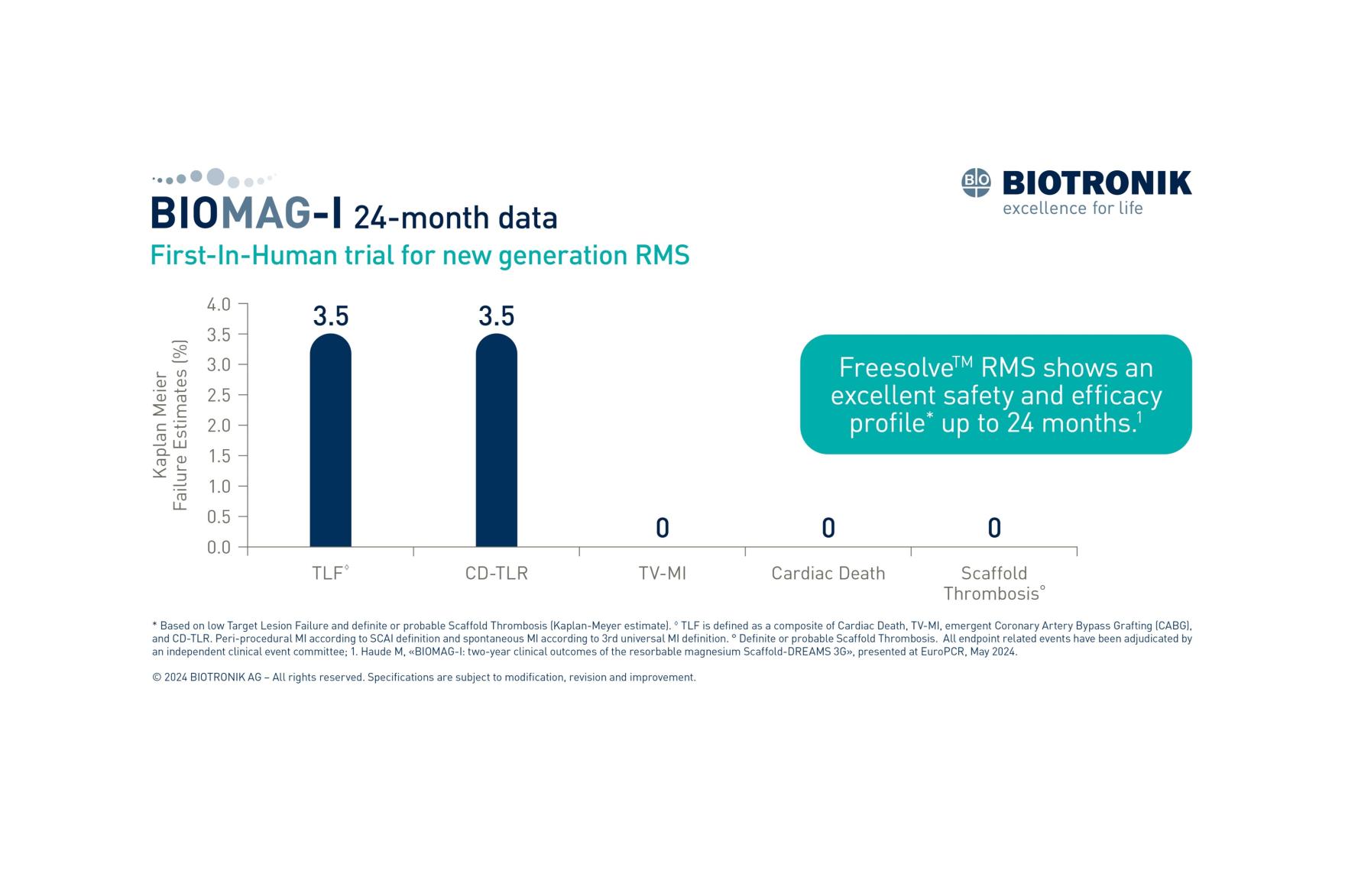
At the 24-month follow-up, the incidence of target lesion failure (TLF) was 3.5% alongside a corresponding 3.5% incidence of target lesion revascularization (TLR) compares favorably with various second-generation DES. No cases of myocardial infarction (MI), cardiac death, or definite/probable scaffold thrombosis were reported.1
“At this two-year time point, the Freesolve RMS with its new Magnesium alloy which allows for increased radial strength, showed again excellent clinical and safety,” said Prof. Michael Haude, Rheinland Klinikum, Neuss, Germany. “With this highly competitive device, we're advancing towards a randomized trial – BIOMAG-II - to compare the advantages of a fully resorbable implant to a permanent DES that remains in the artery indefinitely.”
The initial findings from the BIOMAG-I study 1-year follow-up revealed excellent late lumen loss outcomes at both six and 12 months2,3. Additionally it confirmed that the Freesolve RMS achieves complete absorption* within 12 months post-implantation4 as a result of its proprietary BIOmag® magnesium alloy.
“The new outcomes from the BIOMAG-I trial highlight a very promising future for the era of metallic resorbable magnesium scaffolds,” said Prof. Dr. Georg Nollert, Vice President Medical Affairs, Vascular Intervention at BIOTRONIK. “This strengthens our dedication to innovate and progress through the Freesolve RMS technology, while continuously building strong clinical evidence to ultimately enable the optimal outcomes for patients.”
Additionally, the BIOMAG-II trial has started and is a prospective, international, multi-center randomized-controlled study aiming to evaluate the safety and clinical performance of the new-generation Freesolve RMS, compared to a contemporary DES. The first patient has already been enrolled at the HUG - University Hospital Geneva in Switzerland. The study is primarily evaluating the TLF rate over a 12-month period with consecutive follow-ups.
-END-
References:
1. Haude M., BIOMAG-I: two-year clinical outcomes of the resorbable magnesium Scaffold-DREAMS 3G, presented at EuroPCR 2024
2. Haude M., et al, The Lancet eClinicalMedicine2023;59: 101940
3. Haude M., et al. A new resorbable magnesium scaffold for de novo coronary lesions (DREAMS 3G): one-year results of the BIOMAG-I first.in-human study. EuroIntervention 2023;19
4. Seguchi M., BIOMAG-I: Twelve-months vessel healing profile following the novel resorbable magnesium scaffold implantation: an intravascular OCT analysis of the BIOMAG-I trial, presented at ESC 2023.
*99.3% resorbed at 12 months (markers are not resorbable), based on clinical data.
For more information, please visit: Freesolve RMS
Freesolve™ RMS is available in CE-mark accepting countries.
Freesolve, BIOMAG and Magmaris are trademarks or registered trademarks of the BIOTRONIK Group of Companies.
About BIOTRONIK:
At BIOTRONIK, patient well-being is our top priority and has been for 60 years. BIOTRONIK is a leading global medical technology company with products and services that save and improve the lives of millions suffering from heart and blood vessel diseases as well as chronic pain. Driven by a purpose to perfectly match technology with the human body, we are dedicated innovators who develop trusted cardiovascular, endovascular and neuromodulation solutions. BIOTRONIK is headquartered in Berlin, Germany, and is represented in over 100 countries.