BIOFLOW-V RCT
NCT02389946
Ensaio clínico randomizado que compara o stent com eluição de droga Orsiro1 com o DES Xience2
Conclusão
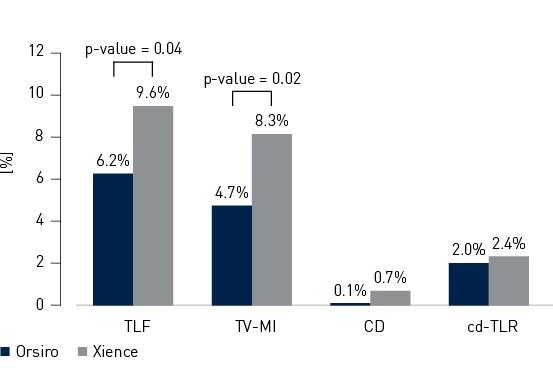
- Neste ensaio clínico internacional randomizado de 1.334 pacientes, Orsiro demonstrou taxas de falha da lesão alvo (TLF) estatisticamente significativamente mais baixas em comparação com Xience em 12 meses (Orsiro 6,2%, Xience 9,6%, valor p = 0,04)
- O sucesso do procedimento foi significativamente maior com Orsiro (Orsiro 93,8%, Xience 90,1%, p-valor = 0,02), principalmente devido a uma maior taxa de enfarte do miocárdio (MI) intra-hospitalar associado a Xience (Orsiro 3,9%, Xience 6,7%, valor p = 0,03)
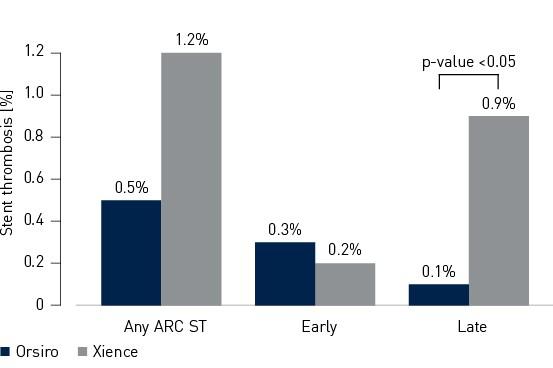
- A taxa de trombose de stent (ST) foi numericamente menor na coorte de Orsiro (Orsiro 0,5%, Xience 1,2%, valor p = 0,18)
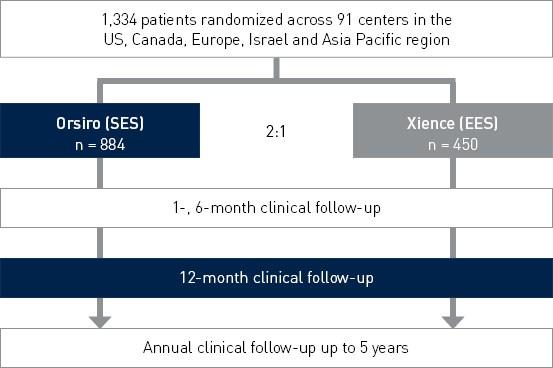
Design de estudo
Ensaio IDE (Investigational Device Exemption), prospectivo, multicêntrico e controlado 2: 1 para avaliar a segurança e eficácia de Orsiro no tratamento de pacientes com até três lesões de novo ou reestenóticas (apenas PTCA padrão).
Investigadores principais:
- Dr. David Kandzari, Piedmont Heart Institute, Atlanta, EUA
- Dr. Jacques Koolen, Catharina Ziekenhuis, Eindhoven, Holanda
Endpoints
Endpoint primário:
RLA em 12 meses definido como um composto de Morte Cardíaca (CD) , Vaso-alvo MI (TV-MI) ou qualquer Revascularização de Lesão Alvo clinicamente orientada (cd-RLA)
Endpoints secundários pré-especificados:
- Componentes do endpoint primário
- Falha da embarcação alvo (TVF) e componentes individuais de TVF
- Morte
- MI e / ou CD
- ST (todos, definido, definido / provável, provável, possível ST) 3
- Taxas de sucesso (dispositivo, lesão e procedimento)
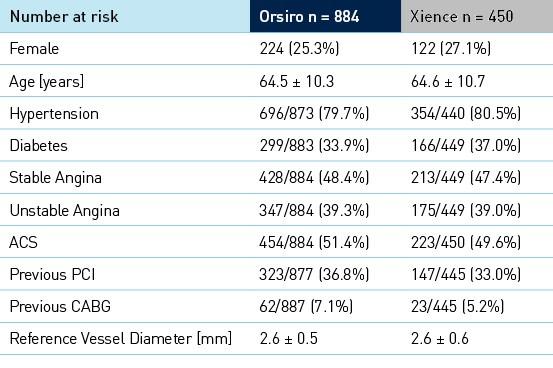
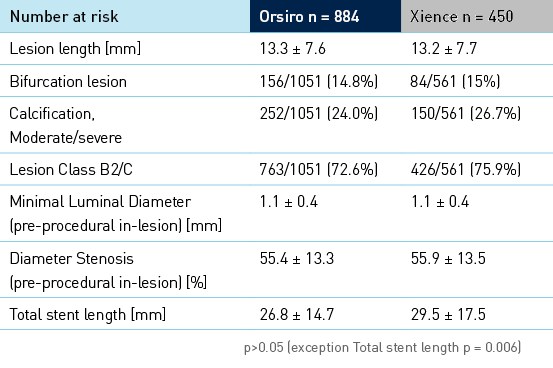
Características clínicas, angiográficas e de procedimento de linha de base
Resultados clínicos
Endpoint primário e compostos em 12 meses
Endpoint secundário - Consórcio de Pesquisa Acadêmica de 12 meses (ARC) ST3
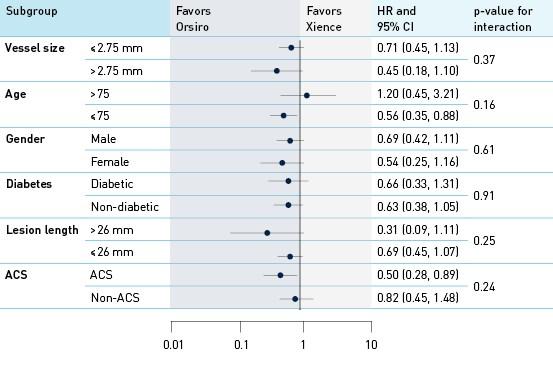
Análise da interação por grupos e subgrupos RLA em 12 meses
Estimativa Kaplan-Meier
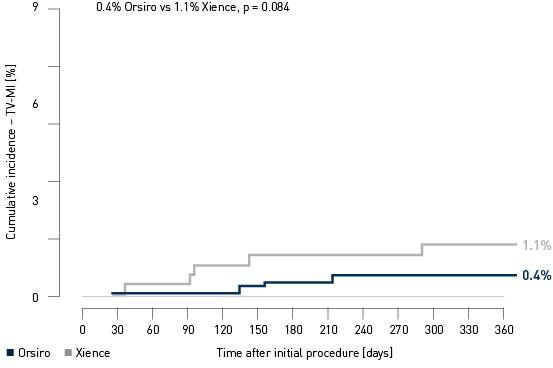
Análise de Marco TV-MI4


Isenção de responsabilidade
© BIOTRONIK AG - Todos os direitos reservados.
As especificações estão sujeitas a modificações, revisões e melhorias.
Fonte
Kandzari D. et al., Ultrathin Biorresorbable Polymer Sirolimus-Eluting Stents versus Thin Durable Polymer-Eluting Stents com Everolimus em pacientes submetidos a revascularização coronária (BIOFLOW-V): um ensaio randomizado, 2017, The Lancet.
1 CUIDADO - Dispositivo investigacional. Limitado pela lei dos Estados Unidos para uso experimental.
3 de acordo com os critérios do Academic Research Consortium (ARC) para trombose de stent aguda, subaguda, tardia, muito tardia e cumulativa
2 Xience é uma marca registrada da Abbott Cardiovascular Systems Inc.
4 Kandzari D. BIOFLOW-V: Um estudo multicêntrico prospectivo randomizado para avaliar a segurança e eficácia do sistema de stent coronário com eluição de Orsiro SiroLimus no tratamento de indivíduos com até três De Novo ou ciência de lesões coronárias restenóticas. Apresentação na ESC 2017.
