SORT OUT VII
NCT01879358
Stent Orsiro com eluição de Sirolimus da BIOTRONIK vs Stent Nobori com eluição de Biolimus da Terumo
Conclusão
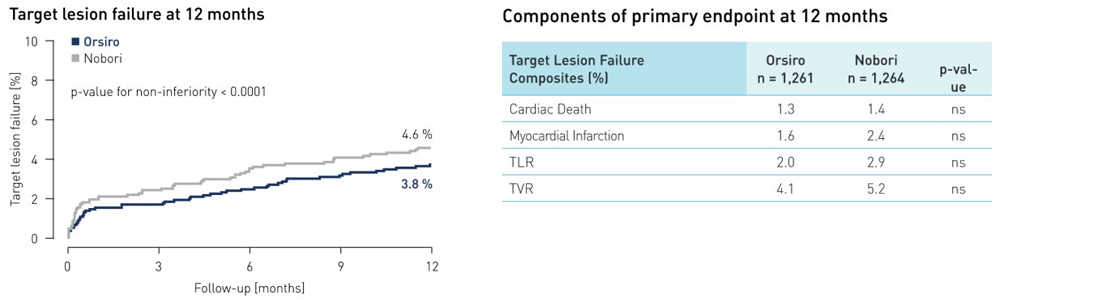
- Dados de um ano de um grande (n = 2.525) ensaio SORT OUT VII independente da indústria apresentado na EuroPCR 2015 demonstram que Orsiro não é inferior ao stent Nobori
- Em um ano, o desfecho primário falha da lesão alvo (TLF) ocorreu em 3,8% dos pacientes Orsiro vs. 4,6% daqueles tratados com SF de Nobori (p não inferioridade <0,0001)
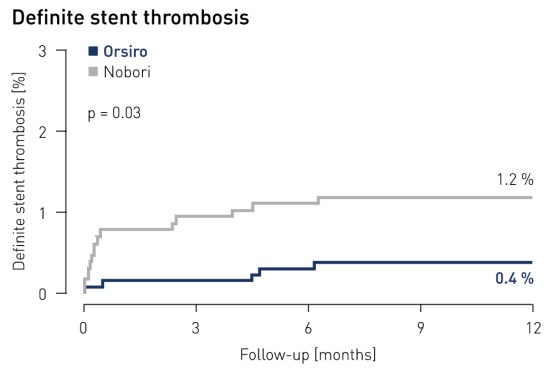
- Os pacientes no braço Orsiro demonstraram uma taxa significativamente mais baixa de trombose de stent definitiva: apenas 0,4% dos pacientes Orsiro em comparação com 1,2% dos pacientes no braço Nobori (p = 0,03)
- Esses resultados altamente encorajadores confirmam os de BIOSCIENCE, a grande comparação independente entre Orsiro e Xience Prime apresentada na ESC 2014 e publicada no The Lancet
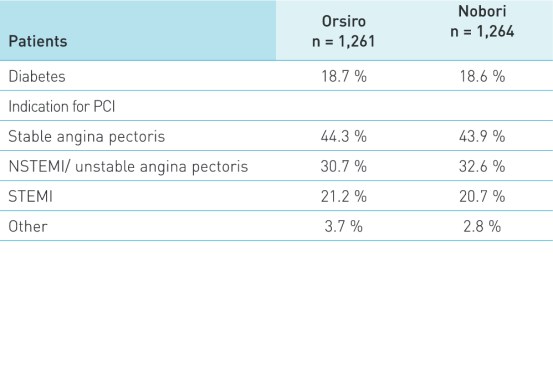
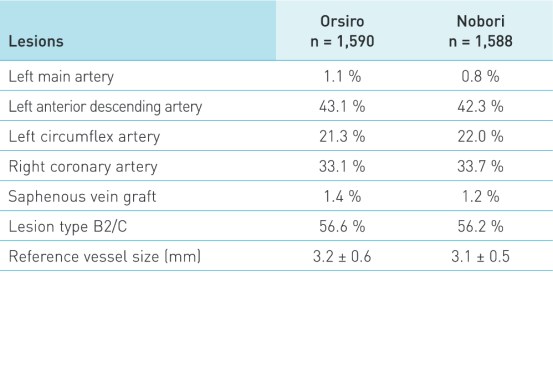
Características do paciente e da lesão
Imagem
Imagem
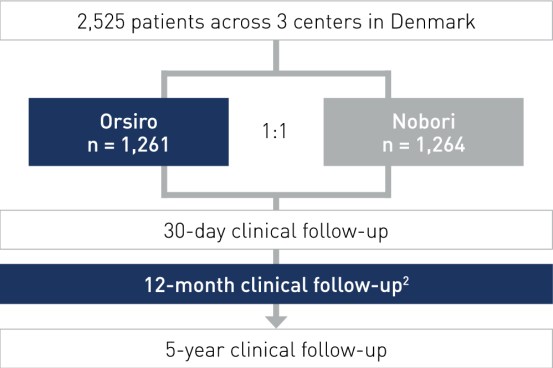
Design de estudo
- All-comers, multicêntrico, randomizado, design de não inferioridade
- Investigadores principais: Prof. Lisette Okkels Jensen, Odense, Dinamarca e Dr. Per Thayssen, Odense, Dinamarca
Imagem
Resultados do endpoint primário
Imagem
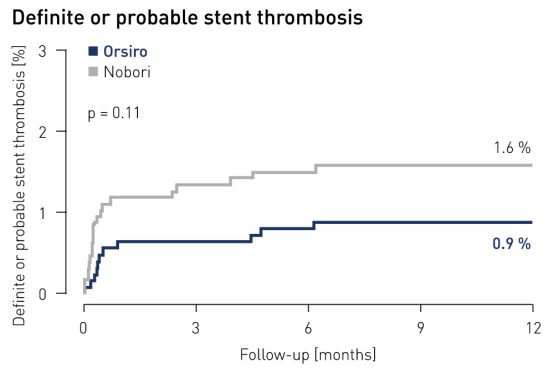
Trombose de stent1 resulta em 12 meses
Imagem
Imagem
Downloads


Fonte
Apresentação, Lisette Okkels Jensen, EuroPCR 2015
Isenção de responsabilidade
© BIOTRONIK AG
Todos os direitos reservados. As especificações estão sujeitas a modificações, revisões e melhorias.
1 Trombose de stent definitiva e provável de acordo com a definição ARC e julgada por um comitê de eventos clínicos independente
2 Endpoint primário: composição de falha da lesão-alvo de morte cardíaca, infarto do miocárdio ou revascularização da lesão-alvo
