PEACE
Eficácia do pulsar - um registro de todos: resultados de 12 meses 1
Conclusão
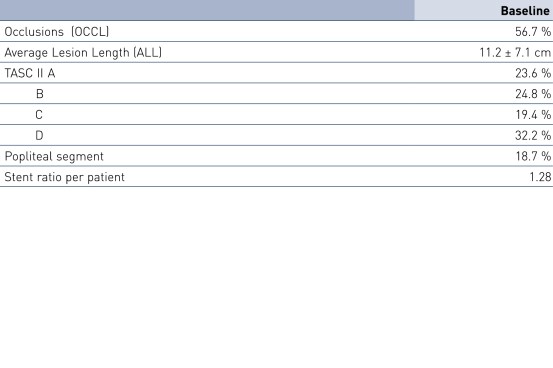
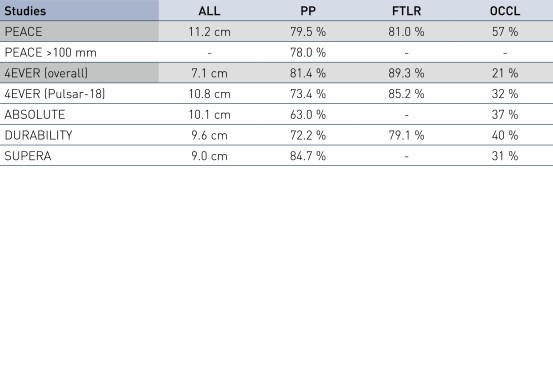
- Os stents pulsares mostraram resultados de estudo favoráveis em 12 meses em pacientes com um comprimento médio de lesão de 11,2 cm
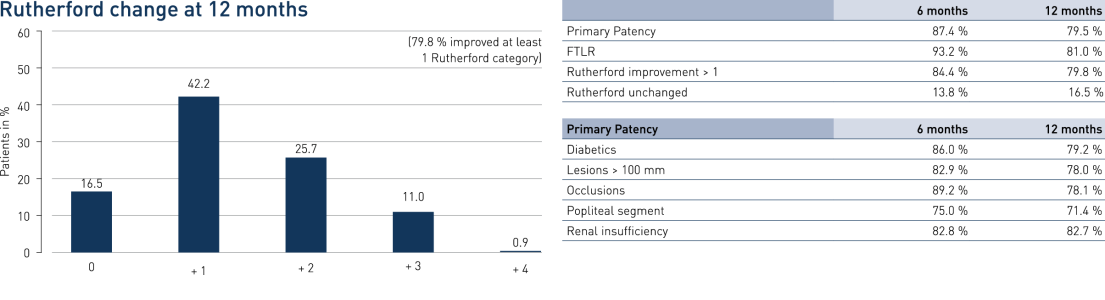
- Uma patência primária (PP) em 12 meses de 79,5% e Liberdade de Revascularização da Lesão Alvo (FTLR) de 81%
- Os resultados são semelhantes aos dados publicados de comprimentos de lesão semelhantes, incluindo 4EVER (Pulsar-18) com um PP de 73,4% e um FTLR de 85,2%
- Nenhuma diferença significativa entre lesões TASC A / B vs. TASC C / D (p = 0,55) e diabéticos vs. não diabéticos (p = 0,92)
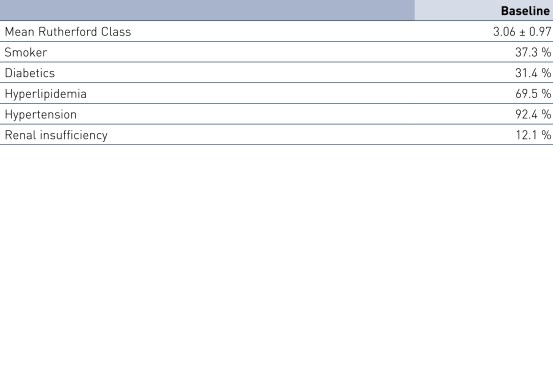
Dados Demográficos de Pacientes
Design de Estudo
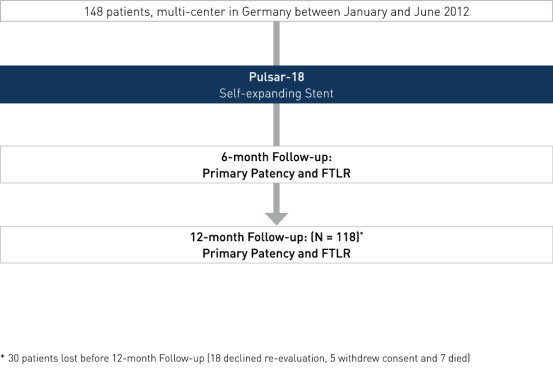
- Registro multicêntrico e potencial de todos os participantes
- Número de pacientes (n): 148 2
- Investigadores primários: Dr. Michael Lichtenberg, Arnsberg, Alemanha e Prof. Dr. Günther Wittenberg, Bielefeld, Alemanha
- Endpoints: PP 3 em 6 e 12 meses e FTLR
- Acompanhamento em 6 e 12 meses
- Centros participantes: Dr. B Hailer, Essen, Alemanha; Dr. Claus Nolte-Ernsting, Mülheim, Alemanha; Dra. Christiane Tiefenbacher, Wesel, Alemanha; Dr. Jawed Arjumand, Wuppertal, Alemanha; Dr. Michael Lichtenberg, Arnsberg, Alemanha; Dr. Günther Wittenberg, Bielefeld, Alemanha
Resultados
PEACE Patência Primária e Liberdade de TLR em Perspectiva
Downloads

INTERVENÇÃO VASCULAR
Liberação de stent de autoexpansão Stent com uma mão para implantação precisa de stent
INTERVENÇÃO VASCULAR
Stent autoexpansível Eixo tri-axial para um sistema de entrega estável durante a implantação do stent
Fontes:
1 Lichtenberg M. JEVT. 2014; 21: 373-380.
2 30 pacientes foram perdidos antes do acompanhamento de 12 meses (18 recusaram a reavaliação, cinco retiraram o consentimento e sete morreram)
3 Definido como ultrassom duplex binário PSVR <2,5 na (s) lesão (ões) alvo com stent e ausência de revascularização da lesão-alvo ( FLR)
EUA: Não está à venda - Investigational Device.
Limitado pela legislação dos Estados Unidos para uso investigativo.
© BIOTRONIK AG
Todos os direitos reservados. As especificações estão sujeitas a modificação, revisão e melhoria.
