QP ExCELs (CRT-DX)
Novel Two-Lead Cardiac Resynchronization Therapy System Provides Equivalent CRT Responses with Less Complications than a Conventional Three-Lead System: Results from the QP ExCELs Lead Registry
Shaik N et al., Journal of Cardiovascular Electrophysiology, May 2020.
doi: 10.1111/jce.14552
Study Design
- Subanalysis of QP ExCELs (prospective, multi-center, observational international registry to evaluate the safety and efficacy of the Sentus QP LV lead in 1907 patients)
- Comparing complication rates and CRT response: two-lead CRT-DX vs. standard CRT-D system
- Including 240 de novo patients with standard CRT indication from 50 U.S. centers (120 pairs matched by gender, NYHA class, and heart failure etiology)
Key Result 1
Significantly Lower Rate of Major Complications with CRT-DX
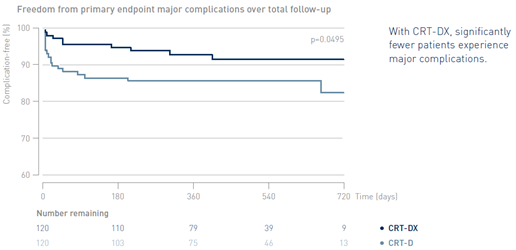
Key Result 2
Lead-Related Major Complications:
Fewer Lead Dislodgements with CRT-DX and Similar Performance of RV Lead
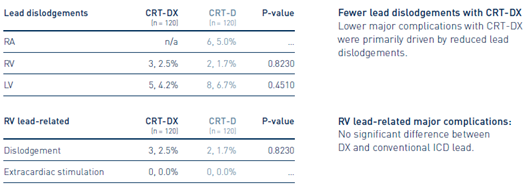
Key Result 3
Similar CRT Responses Shown in LV Pacing and Clinical Outcome Parameters
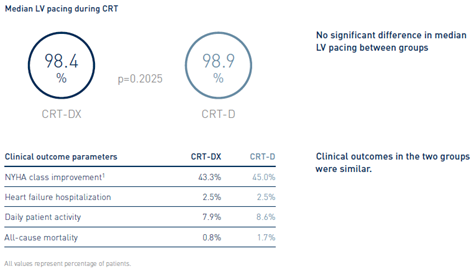
Clinical Relevance
- First study to compare major complications in CRT-DX systems with standard CRT-D systems in a multi-center, real-life registry
- The results show that the CRT-DX system can provide similar CRT responses and significantly fewer complications, indicating that the CRT-DX system is a capable alternative in patients without an atrial pacing indication.2
2 Author’s conclusion extracted from publication.
| Study Objective |
|
|---|---|
| Primary Endpoint |
|
| Additional Data of Interest |
CRT device performance and response:
|
| Clinical Sites |
|
| Sample Size |
|
| Main Inclusion Criteria |
|
| Study Flowchart | 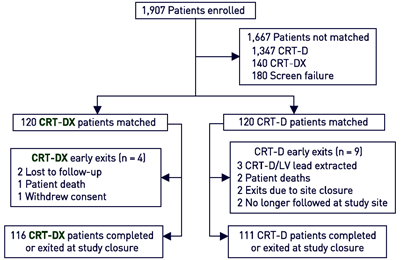 |
| Follow-Up |
Mean follow-up
|
| Study Duration |
|
| Reference no. |
|
